A new multi-centre study from the UK offers promising real-world evidence for patients battling advanced oesophago-gastric (OG) cancers, confirming that modern chemoimmunotherapy regimens deliver survival outcomes comparable to those seen in landmark clinical trials.
For patients diagnosed with advanced or metastatic oesophageal, gastro-oesophageal junction (GOJ), or gastric cancers, the prognosis has historically been grim. With five-year survival rates often hovering between 18% and 23.9%, and dropping to as low as 4–10% for metastatic disease, the medical community has long sought more effective frontline interventions.
A significant shift occurred with the advent of immune checkpoint inhibitors like pembrolizumab and nivolumab. When combined with traditional platinum- and fluoropyrimidine-based chemotherapy, these drugs have demonstrated life-extending potential in phase III trials. However, a lingering question remained: do these benefits translate to the heterogeneous, often more frail population seen in routine NHS practice? A new study published in the journal Cancers (2026) suggests that the answer is a resounding yes.

Main Facts: Bridging the Gap Between Trials and Practice
The retrospective study, led by researchers at The Clatterbridge Cancer Centre and Velindre Cancer Centre, analyzed 76 patients who received first-line palliative chemoimmunotherapy between April 2021 and July 2024. The data provides a crucial "real-world" mirror to the highly controlled environments of clinical trials like KEYNOTE-590 and CheckMate-649.
Key findings include:
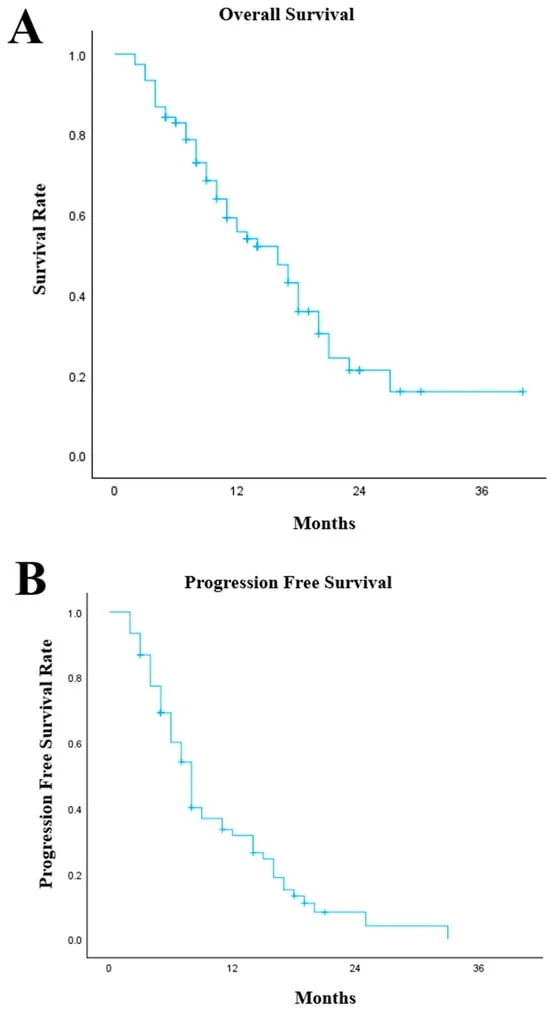
- Overall Survival (OS): The median survival for the cohort reached 16 months, a figure that stands toe-to-toe with the 12–14 month outcomes reported in pivotal international trials.
- Progression-Free Survival (PFS): Patients experienced a median PFS of 8 months, indicating a robust period of disease control before the next line of therapy was required.
- Disease Control: An impressive 80.3% of patients achieved some form of disease control (complete response, partial response, or stable disease).
- Manageable Toxicity: While immune-related adverse events (irAEs) were documented in 40.8% of patients, the vast majority were low-grade (Grade 1–2). Severe toxicities (Grade 3 or higher) occurred in 13.2% of the group, with two treatment-related deaths recorded.
Chronology of the Research
The study’s timeline reflects the rapid adoption of new standards of care within the UK National Health Service (NHS) following approval by the National Institute for Health and Care Excellence (NICE).

- April 2021: Commencement of the observation period. This marked the implementation of chemoimmunotherapy as a standard first-line palliative treatment for eligible patients in the participating centres.
- 2021–2024: The data collection phase. During this period, 76 patients were identified and tracked through electronic health records. Researchers documented demographic characteristics, histological subtypes, baseline inflammatory markers, and radiological response to treatment.
- March 2026: The study was formally submitted for peer review.
- May 2026: Following a rigorous revision process, the article was accepted and subsequently published on 9 May 2026, providing a definitive look at three years of clinical practice.
Supporting Data: Understanding the Patient Profile
The cohort was representative of the complex clinical reality faced by oncologists. The majority of patients (59.2%) were aged 65 or older, and 71.1% presented with metastatic (Stage IV) disease. The study population was heavily skewed toward adenocarcinoma (81.6%), with 18.4% presenting with squamous cell carcinoma.
One of the most compelling aspects of the data is the breakdown of survival by treatment response. Patients who achieved disease control saw their median OS jump to 17 months, compared to just 4 months for those whose disease progressed immediately. While researchers caution that this could be influenced by "guarantee-time bias"—the idea that patients must live long enough to be scanned to be counted as "responders"—it underscores the profound clinical benefit for those who are sensitive to the immunotherapy.
The research also delved into exploratory biomarkers. While not reaching statistical significance, the study noted that higher baseline lymphocyte counts and lower neutrophil-to-lymphocyte ratios (NLR) were associated with better survival outcomes. These markers may eventually help clinicians predict which patients are most likely to respond, though the authors emphasize that these results are currently hypothesis-generating.

Official Responses and Clinical Interpretation
The authors of the study, including lead investigator James Birch-Ford and senior contributors from Clatterbridge and Velindre, frame these findings as a strong endorsement of current NICE guidelines. "In this real-world UK multi-centre cohort, first-line chemoimmunotherapy demonstrated survival outcomes comparable to pivotal clinical trials, with manageable toxicity," the team concluded.
They highlight that while clinical trials are essential for drug approval, they often exclude the "real-world" patient—those with lower performance statuses, multiple comorbidities, and varying levels of fitness. The fact that the 16-month survival figure matches or slightly exceeds the trial benchmarks is particularly encouraging for clinicians who may have been concerned about the translation of trial data to their daily practice.
However, the team maintains a cautious stance on toxicity management. Despite the manageable profile of the drugs, the study documented a range of immune-mediated conditions, including hepatitis, colitis, and rare but serious instances of meningoencephalitis. The message to the clinical community is clear: immunotherapy is not "chemo-lite." It requires rigorous, multidisciplinary oversight to identify and treat immune-related adverse events before they become life-threatening.

Implications for Future Care
What does this mean for the future of oncology in the UK? The researchers suggest that the next frontier is not just proving that these treatments work, but understanding for whom they work best.
1. The Need for Prospective Registries
The retrospective nature of the study, while valuable, carries the inherent risks of selection bias and missing data. The authors advocate for prospective, multi-centre collaborative studies. By building a larger, standardized registry of patients, the medical community could move beyond exploratory biomarkers like NLR and develop a robust "predictive signature" for patient selection.
2. Refining Patient Selection
With PD-L1 expression levels currently used to guide eligibility under NICE criteria, the study suggests that we are only scratching the surface of biological prediction. Future research will likely focus on integrating genomic profiling and detailed immune-environment mapping to refine treatment pathways.

3. Optimization of Toxicity Management
The study reinforces the "long tail" effect of immunotherapy—some patients achieve durable, long-term responses. The authors emphasize that keeping patients on therapy is the priority. Therefore, developing standardized, rapid-response protocols for managing low-grade toxicities is as critical as the drug regimen itself.
4. Broadening Access
Finally, the findings suggest that the benefits of chemoimmunotherapy are consistent across different primary tumor sites (oesophageal, GOJ, and gastric). As these treatments become more deeply embedded in the NHS, the focus will shift toward ensuring equitable access across all tertiary centers, ensuring that every eligible patient has the opportunity to achieve the survival outcomes documented in this study.
In conclusion, the study serves as a vital bridge between the abstract success of international drug trials and the concrete, day-to-day work of UK cancer centers. By confirming that real-world outcomes can rival those in clinical trials, the research provides both clinicians and patients with renewed optimism for managing what has historically been one of the most challenging groups of malignancies to treat. As the field moves toward more personalized medicine, the lessons learned from this 76-patient cohort will undoubtedly inform the next generation of clinical practice.