In recent years, the oncology community has been confronted with a sobering, paradoxical trend: while traditional behavioral risk factors for cancer—such as tobacco use and poor diet—have largely plateaued or declined in developed nations, the incidence of cancer among younger adults is steadily climbing. Two landmark studies released in the United Kingdom have now shed new light on this phenomenon, pointing to obesity as a significant, yet enigmatic, driver of this health crisis. These findings not only challenge our current understanding of how we measure health in cancer patients but also demand a fundamental shift in how public health policies approach cancer prevention.
Main Facts: A New Paradigm for Cancer Etiology
The emerging evidence suggests that the relationship between excess body weight and cancer is far more intricate than previously understood. According to the research, obesity is no longer just a potential precursor to health issues in later life; it is increasingly a primary factor in the development of malignancies among those under the age of 50.
The primary takeaway from the recent literature is twofold. First, excess weight is unequivocally a strong contributor to the surge in early-onset cancer rates, particularly in the UK. Second, current diagnostic and research methods, which rely heavily on single, point-in-time Body Mass Index (BMI) measurements, are fundamentally flawed. They fail to account for "lifetime obesity exposure," effectively masking the true cumulative impact that weight has on biological systems over decades. By failing to track a patient’s weight history, clinicians and researchers are likely underestimating the degree to which obesity fuels the cancer epidemic.
Chronology: Tracing the Rise of the "New" Cancer Epidemic
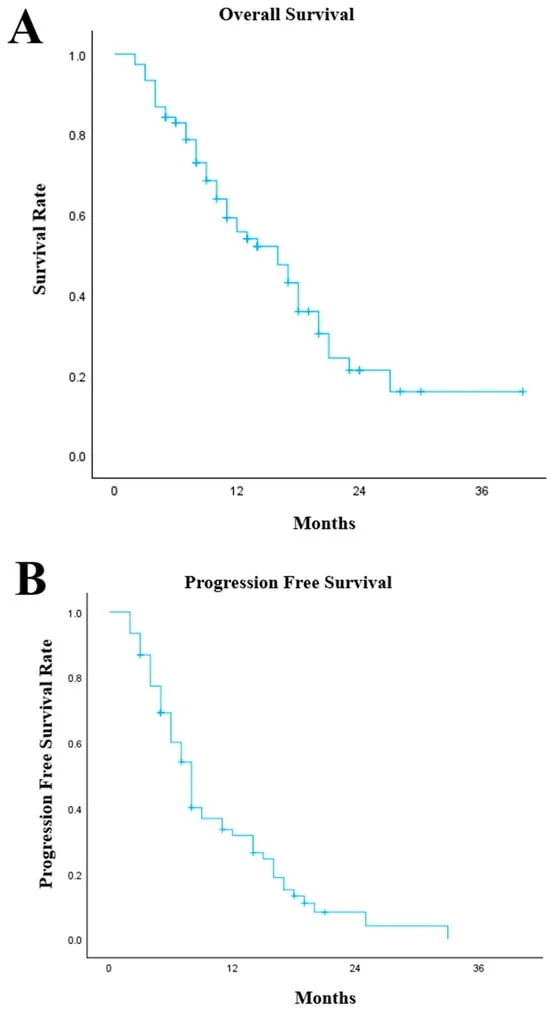
To understand the current landscape, researchers looked back at decades of registry data. By analyzing national cancer registry records spanning from 2001 to 2019, scientists at The Institute of Cancer Research (ICR) and Imperial College London sought to map the rise of 22 different types of cancer in women and 21 in men.
- 1995–Present: This period marked a consistent, steady rise in overweight and obesity rates across the UK population. Researchers noted that the steepest increases were recorded in younger women, with a 2.6% relative increase in obesity prevalence per year.
- 2001–2019: During these nearly two decades, the study analyzed the incidence of 11 specific cancers known to be linked to behavioral factors. Notably, as obesity rates climbed, other traditional risk factors—smoking, alcohol consumption, high intake of processed meats, and sedentary behavior—remained stagnant or even saw downward trends.
- The Discrepancy: The timeline reveals a widening gap. While obesity rates moved in tandem with cancer rates, the researchers discovered that the statistical increase in cancer cases could not be explained by BMI shifts alone. For instance, in bowel cancer cases among younger women, only a portion of the rise could be mathematically attributed to BMI increases, indicating that unknown "early-life exposures" or environmental shifts are working in tandem with weight gain.
Supporting Data: When Current Weight Lies
Perhaps the most startling revelation comes from a large-scale real-world study led by the University of Oxford, which scrutinized the medical histories of over 79,000 patients across 13 different cancer types. The data exposes the "snapshot bias" of current clinical assessments.
The study found that in every one of the 13 cancer types studied, the lifetime prevalence of obesity exceeded 50%. The most striking example was found in pancreatic cancer: at the time of treatment initiation, only 13.7% of patients were classified as obese by standard BMI metrics. However, when the researchers dug deeper into their longitudinal health records, they discovered that 55.8% of those same patients had experienced obesity at some point in their lives.
This data point is revolutionary. It suggests that the biological "insult" of obesity—chronic inflammation, hormonal imbalances, and metabolic disruption—is cumulative. A patient who is a healthy weight at age 50 may still be at higher risk for cancer if they lived with obesity in their 20s or 30s. This "legacy effect" of weight remains largely invisible to doctors who only look at the weight of a patient on the day they walk into an oncology ward.
Official Responses and Expert Analysis
The scientific community has reacted to these findings with a mix of urgency and calls for systemic change. Montserrat García-Closas of The Institute of Cancer Research, a lead author on the multi-institutional study published in BMJ Oncology, emphasized that the data points toward a complex, multi-factorial cause.
"Our findings show that while cancer rates are rising in younger adults, the trends are unlikely to be explained by changes in most known behavioral risk factors," García-Closas stated. "Excess weight is an important contributor, although it cannot fully account for the scale of the rise in bowel and other cancers. This tells us that multiple factors—including early-life exposures—may be acting together."
Dr. Simon Lord of the University of Oxford, who led the research on lifetime obesity exposure, highlighted the clinical implications. "This work highlights the limitations of using a single BMI measurement, which fails to accurately reflect past obesity exposure," Lord noted. "How obesity affects cancer prognosis is extremely complex, with both current and previous obesity likely to be important."
Both research teams agree that the medical establishment must pivot away from "snapshot" diagnostics and toward longitudinal data tracking. For chemotherapy patients, this is particularly vital, as dosing calculations are often based on current weight—a metric that may be misleading if the patient’s metabolic history is ignored.
Implications for Clinical Care and Public Health
The convergence of these studies presents a clear mandate for both clinicians and policymakers. The implications can be categorized into three primary domains:
1. Clinical Practice: Rethinking Patient Assessment
Oncologists should incorporate "obesity history" into the standard intake process. By reviewing longitudinal BMI data, clinicians can better predict treatment outcomes, manage toxicities in chemotherapy, and identify patients who might require more aggressive monitoring for secondary malignancies.
2. Public Health: A Shift to Early Intervention
The finding that younger populations are experiencing a surge in cancer rates linked to obesity demands a proactive approach. Prevention cannot wait until middle age. Stronger public health policies, including improved access to nutritional education, restrictions on the marketing of ultra-processed foods, and the creation of built environments that encourage physical activity, are essential for children and young adults. As García-Closas asserted, "Tackling obesity across all ages… could slow the rise in cancer and prevent many cancers—and must become a national priority."
3. Future Research: The Search for the "Missing" Factors
While obesity is clearly a central player, the data confirms it is not the sole culprit. The fact that non-BMI-attributable cancer rates are rising suggests that scientists must now look toward other environmental or developmental factors. Potential areas for investigation include the role of the microbiome, environmental pollutants (endocrine disruptors), and the long-term impact of dietary patterns established in childhood.
Conclusion: A Call to Action
The evidence is clear: the rise in cancer rates among the under-50 demographic is not merely a statistical anomaly or a byproduct of better screening. It is a reflection of a changing biological landscape, heavily influenced by the global obesity crisis. With global projections suggesting that over 2 million cancer cases could be attributable to obesity by 2070, the window for effective intervention is closing.
By recognizing that obesity is a lifetime journey rather than a single data point, the medical community can move toward a more nuanced, effective, and patient-centered model of care. The task ahead is immense, requiring a transition from reactive cancer treatment to a comprehensive, life-course approach to metabolic health and cancer prevention. The future of oncology, it seems, depends on our ability to address not just the cancer itself, but the historical weight of the patient’s life.