The management of cancer treatment is a delicate balancing act. While clinicians strive to maintain the intensity and schedule of chemotherapy regimens to maximize oncological outcomes, they are frequently hamstrung by the physiological limitations of their patients. Among the most persistent barriers to consistent care is chemotherapy-induced thrombocytopenia (CIT)—a condition characterized by a significant drop in platelet counts that leaves patients vulnerable to bleeding and necessitates disruptive treatment delays or dose reductions.
Recent findings from the Phase III RECITE trial have provided a potential paradigm shift in this landscape, highlighting the efficacy of romiplostim in maintaining treatment continuity for patients with gastrointestinal (GI) cancers. To better understand the clinical implications of these findings, we sat down with Dr. Gerald Soff of the Sylvester Comprehensive Cancer Center at the University of Miami Miller School of Medicine.
The Clinical Challenge: Why CIT Matters
In the context of gastrointestinal cancers—such as colorectal, pancreatic, and gastric malignancies—the therapeutic window is often narrow. The efficacy of these regimens depends heavily on dose intensity. When a patient’s platelet count drops, the clinical decision-making process becomes fraught with compromise.
"The major adverse impact of CIT is the forced modification of treatment plans," explains Dr. Soff. "When a patient develops thrombocytopenia, we are often left with a binary choice: either delay the next cycle of chemotherapy or reduce the dose. Both options are suboptimal. A delay allows the tumor a ‘breather’ to potentially develop resistance or progress, while a dose reduction inherently limits the cytotoxic power of the treatment we are trying to deliver."
For patients already fighting aggressive GI cancers, these interruptions are not merely logistical inconveniences; they are setbacks that can fundamentally alter the prognosis. The clinical goal, therefore, is to support the bone marrow in a way that allows the patient to tolerate the full planned dose of chemotherapy on time.
Chronology of the RECITE Trial: A New Standard of Care
The development of thrombopoietin receptor agonists (TPO-RAs) like romiplostim has evolved over the last decade, transitioning from the treatment of benign conditions like immune thrombocytopenia (ITP) to more complex supportive care roles in oncology.
Phase I and II Foundations
Initial studies focused on the safety profile of TPO-RAs in cancer patients. Early data suggested that these agents could effectively stimulate megakaryopoiesis—the production of platelets—without causing adverse interactions with common chemotherapeutic agents. These pilot studies established the dosing thresholds and confirmed that romiplostim could be administered safely in conjunction with various GI-specific chemotherapy protocols.
The RECITE Trial Design
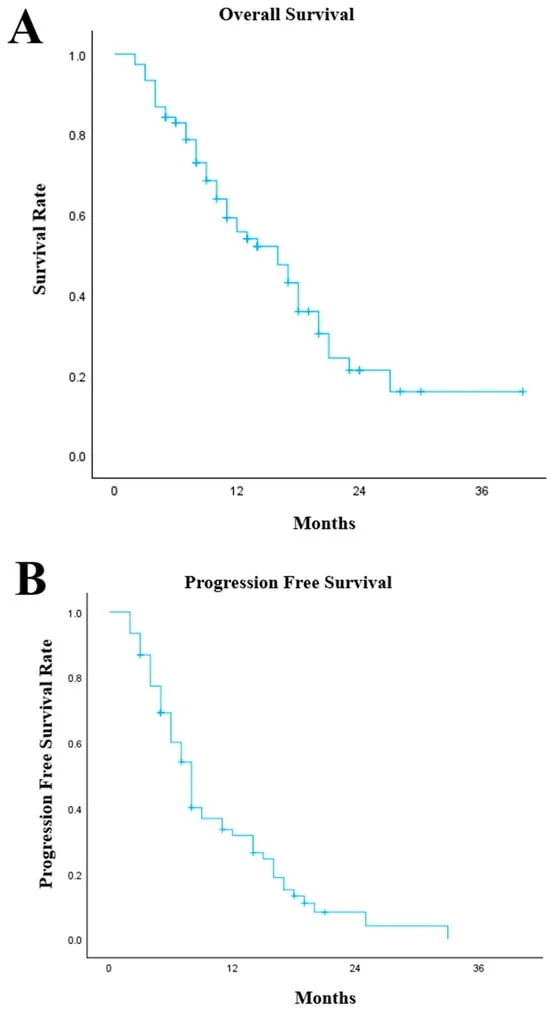
The Phase III RECITE trial was designed to rigorously test the hypothesis that proactive use of romiplostim could prevent the cycle delays and dose reductions caused by CIT. The study enrolled a diverse cohort of patients undergoing myelosuppressive chemotherapy for advanced GI malignancies. The primary endpoint was the successful completion of chemotherapy cycles without the need for unplanned dose modifications or significant treatment delays due to low platelet counts.
Recent Data Presentation
The data released from the RECITE trial earlier this year demonstrated a statistically significant improvement in treatment adherence among the romiplostim cohort compared to the placebo group. The results suggest that by maintaining platelet counts within a safer range, clinicians were able to keep patients on their intended dose-intensity schedules, thereby adhering more strictly to the gold-standard treatment guidelines.
Supporting Data: Examining the Mechanism
Romiplostim functions by binding to and activating the thrombopoietin receptor, mimicking the natural ligand that stimulates the production of platelets in the bone marrow. In the context of chemotherapy, which often suppresses the bone marrow’s innate ability to produce these cells, romiplostim acts as an "accelerator."
Key Trial Metrics
- Treatment Continuity: Patients receiving romiplostim showed a higher percentage of cycles completed without delay compared to those in the control arm.
- Dose Intensity: The cumulative dose of chemotherapy delivered over the course of the study was higher in the romiplostim group, suggesting that the drug effectively mitigated the need for therapeutic tapering.
- Safety Profile: Adverse events associated with romiplostim were consistent with previous findings in other patient populations, with a low incidence of thromboembolic events, which remains a primary concern for clinicians managing patients with cancer.
Expert Perspectives: Dr. Gerald Soff on Real-World Application
In practice, the transition from trial data to the clinic is rarely seamless. Dr. Soff emphasizes that managing CIT requires a personalized approach.
"When we talk about the ‘real-world’ setting, we are talking about patients with significant comorbidities, age-related vulnerabilities, and varying levels of disease burden," Dr. Soff notes. "The RECITE trial gives us a tool, but the clinician must still exercise judgment."
Practical Strategies for the Clinic
Dr. Soff outlines three core pillars for managing CIT:
- Early Risk Stratification: "We identify patients who are at high risk of CIT before they start their first cycle. If we know a regimen is particularly myelosuppressive, we have a lower threshold for starting supportive agents."
- Continuous Monitoring: "Platelet counts aren’t just numbers on a chart; they are the gatekeepers of our treatment. We utilize frequent monitoring to predict a decline before it reaches a critical, cycle-delaying nadir."
- Collaborative Care: "Managing thrombocytopenia requires a multidisciplinary team, including hematologists and oncologists, to ensure that the supportive care intervention is timed perfectly with the chemotherapy schedule."
Implications for Future Oncology Practice
The implications of the RECITE trial extend beyond the specific use of romiplostim. It signals a broader shift toward "proactive supportive care." Historically, oncology has been reactive—we treat the side effects of chemotherapy as they arise. The data from RECITE supports a more preventative philosophy.
Impact on Treatment Outcomes
By preventing delays, clinicians can potentially improve long-term survival outcomes. In many GI cancers, the "window of opportunity" for curative or life-prolonging chemotherapy is finite. Reducing the time spent waiting for blood counts to recover means more cycles of chemotherapy are delivered within that critical timeframe.
The Economic and Patient-Centered View
Beyond clinical efficacy, there is the patient experience. Frequent blood draws, clinic visits for platelet transfusions, and the anxiety associated with treatment delays take a toll on the quality of life. An agent that simplifies the treatment schedule can reduce the burden on the patient and the healthcare system.
Official Responses and Industry Outlook
The oncology community has met the RECITE findings with cautious optimism. Major cancer organizations and oncology nursing societies are currently reviewing the data to determine if clinical practice guidelines should be updated.
"The medical community is always looking for ways to improve the patient journey," says Dr. Soff. "While we still need to collect long-term data on the survivorship outcomes of these patients, the immediate benefit of keeping a patient on their treatment track is undeniable. If romiplostim becomes a standard of care for CIT in GI malignancies, we could see a significant change in how we view the ‘tolerability’ of aggressive chemotherapy."
Looking Ahead
The next phase of research will likely involve investigating the cost-effectiveness of these treatments and identifying specific biomarkers that can predict which patients will benefit most from TPO-RA therapy. As precision medicine continues to evolve, the ability to tailor supportive care as carefully as the primary cancer treatment will be the hallmark of the next generation of oncological practice.
Conclusion
Chemotherapy-induced thrombocytopenia remains a formidable adversary in the treatment of gastrointestinal cancers. However, the data provided by the RECITE trial offers a promising pathway toward better control and improved patient outcomes. By bridging the gap between potent chemotherapy and bone marrow support, clinicians like Dr. Soff are gaining the tools necessary to ensure that patients receive the care they need, on the schedule they deserve.
As we move forward, the integration of agents like romiplostim into standard protocols will require careful navigation, balancing the benefits of treatment continuity against the complexities of individualized patient needs. Nevertheless, the trajectory of current research suggests a future where treatment delays due to manageable side effects become the exception rather than the rule.