Executive Summary: A Resilient Financial Outlook
In a defining moment for the global metabolic health sector, Danish pharmaceutical titan Novo Nordisk has signaled a significant shift in its financial trajectory. In its latest earnings report, the company announced that its sales and profit forecasts for the upcoming fiscal periods are expected to contract less severely than previously anticipated. This upward revision in market expectations is primarily attributed to the explosive early adoption of its new oral version of Wegovy, a development that has provided a much-needed morale boost to the organization following a tumultuous year of market turbulence and intense competition from Eli Lilly.
For investors and analysts, the news provides a window into a pharmaceutical landscape currently defined by the race for GLP-1 (glucagon-like peptide-1) dominance. While Novo Nordisk has faced significant operational headwinds—including sweeping leadership changes, a massive restructuring plan involving the layoffs of 9,000 employees, and the loss of its pole position in the obesity drug market to Eli Lilly’s Zepbound—the rapid uptake of its oral Wegovy pill offers a tangible pathway to reclaiming its status as the market leader.
The Chronology of a Competitive Tug-of-War
To understand the current significance of the oral Wegovy pill, one must view it within the context of the last 18 months of aggressive maneuvering between Novo Nordisk and Eli Lilly.
- The Struggle for Supremacy (2024): After enjoying an early-mover advantage with the injectable version of Wegovy, Novo Nordisk struggled to keep pace with the clinical and commercial momentum of Eli Lilly’s Zepbound. The disparity in supply chain reliability and the efficacy profile of Lilly’s drug began to erode Novo’s market share.
- Organizational Overhaul (Late 2024–Early 2025): Recognizing the existential threat posed by these market shifts, Novo Nordisk initiated a radical internal transformation. This included the departure of key executives, the installation of new leadership, and a painful, large-scale restructuring program designed to streamline the company’s focus on high-growth R&D.
- The Counter-Offensive (Q1 2026): Novo Nordisk launched the oral version of Wegovy on January 5, 2026, marking a pivotal moment in its strategy to move away from the logistical hurdles of injectable pens.
- The Response (Q2 2026): Eli Lilly introduced its own oral competitor, Foundayo, on April 9, 2026. This introduction marked the beginning of a direct, head-to-head battle for the burgeoning "pill-form" segment of the anti-obesity market.
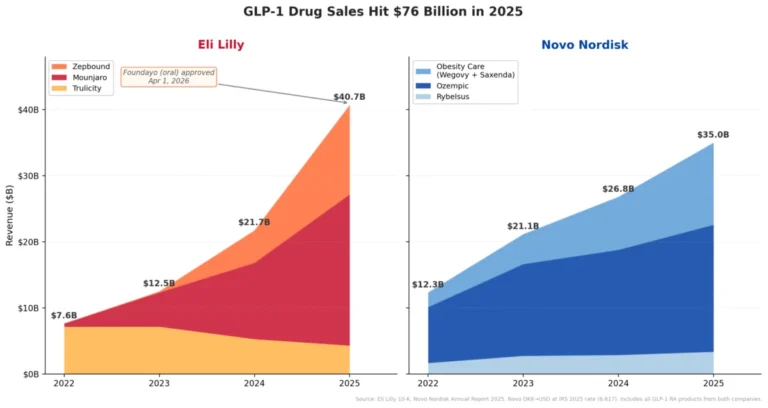
Supporting Data: By the Numbers
The current data indicates that while the market is vast, consumer preference—at least in the early stages—is skewing heavily toward the Novo Nordisk offering.
The Prescription Gap
As of the latest reporting period, the disparity in adoption rates is stark. Novo Nordisk reported that more than 2 million prescriptions have been written for the Wegovy pill since its January launch. During its first full week on the market, the drug recorded over 18,000 prescriptions.

In contrast, Eli Lilly’s Foundayo, which launched in April, recorded 5,612 prescriptions in its third week of availability. Industry analysts note that while the time-to-market difference explains part of this gap, the velocity of the Wegovy pill’s adoption suggests that Novo’s brand recognition remains a potent force in the pharmacy setting.
Clinical Efficacy and Patient Experience
The debate between the two pharmaceutical giants is not merely about volume; it is about clinical performance. While a direct head-to-head clinical trial has not been conducted to compare the oral versions, data from individual clinical trials suggest that patients using the Wegovy pill achieved a higher percentage of total body weight loss compared to those in trials for Lilly’s Foundayo.
Eli Lilly, however, maintains a key competitive advantage in patient experience: Foundayo does not require the strict "empty stomach" fasting protocols associated with some other oral GLP-1 medications. This ease of use remains the cornerstone of Lilly’s marketing strategy as they attempt to close the prescription volume gap.
Official Responses and Strategic Pivot
The leadership at both companies has been vocal about the importance of these oral therapies. Novo Nordisk’s leadership, including CEO Mike Doustdar, has characterized the oral pill as the company’s "best shot" at clawing back lost market share. By offering the oral version alongside a high-dose injectable at a more competitive price point than Lilly’s highest-dose Zepbound, Novo is attempting to create a "pincer movement" on the market, targeting both patients who prefer the convenience of a pill and those who require the potent efficacy of an injectable.
Eli Lilly CEO David Ricks has offered a measured perspective. In recent comments to CNBC, Ricks emphasized that because Foundayo is an entirely new chemical entity—unlike the oral Wegovy, which is an oral formulation of a long-established injectable molecule—it may require a longer runway to build a durable prescription base. "We are in the early innings," Ricks noted, suggesting that the initial prescription disparity is a temporary artifact of brand familiarity rather than a reflection of long-term commercial viability.

Strategic Implications: The Innovation Arms Race
The success of the Wegovy pill is not just a commercial win; it is a tactical necessity for Novo Nordisk, which has faced significant R&D setbacks elsewhere.
The CagriSema Challenge
Novo’s attempt to introduce a next-generation "combination shot" known as CagriSema encountered a major hurdle when clinical results released in February 2026 showed it failed to outperform Eli Lilly’s Zepbound in head-to-head metrics. This failure placed immense pressure on the company to ensure that its oral pipeline succeeded.
The Triple-Acting Horizon
While Novo focuses on stabilizing its current portfolio, the future of the obesity market is shifting toward "triple-acting" drugs. Eli Lilly has reported strong success in late-stage testing of its triple-acting drug, which has shown efficacy not only in diabetes and weight loss but also in treating osteoarthritis. Novo Nordisk is currently in the process of attempting to catch up with its own triple-G agonist research, but industry analysts generally agree that they are currently trailing behind Lilly’s developmental timeline.
Conclusion: The Road Ahead
The market for obesity treatments is projected to reach unprecedented valuations by the end of the decade, yet the "winner-takes-all" dynamic remains elusive. For Novo Nordisk, the strong performance of the Wegovy pill serves as a critical buffer. It buys the company time to refine its R&D pipeline and navigate the structural changes initiated by its leadership.
However, the pharmaceutical industry is notoriously unforgiving. The "pill wars" have only just begun. As Eli Lilly refines its distribution for Foundayo and continues to iterate on its triple-acting compounds, the competitive pressure will only intensify. For patients, the rivalry between these two behemoths is yielding rapid innovation and, eventually, a greater variety of treatment modalities. For the market, the coming year will serve as the ultimate test of whether Novo Nordisk’s strategic pivot toward oral medication is enough to secure its long-term dominance in a sector that is, quite literally, reshaping the world’s approach to metabolic health.
Key Takeaways for Stakeholders:
- Financial Resilience: Novo Nordisk’s earnings indicate a successful containment of the "Lilly effect" via the Wegovy pill.
- Market Adoption: The 2-million-prescription milestone for the Wegovy pill underscores the massive consumer demand for non-injectable options.
- The Innovation Gap: While Novo is winning on current volume, Eli Lilly’s progress with triple-acting drugs suggests that the next phase of the competition will be won in the laboratory, not just at the pharmacy counter.
- Operational Stability: Despite the massive 9,000-person layoff, the company’s ability to deliver consistent results suggests that the organizational restructuring is having the desired effect of sharpening operational efficiency.