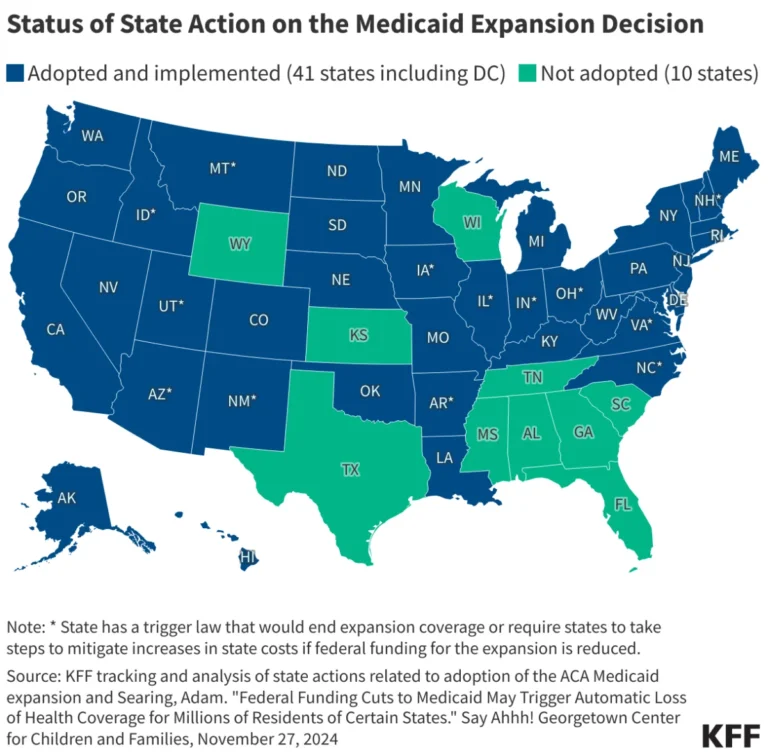
The development of therapies for ultra-rare diseases—conditions affecting fewer than 1,000, and sometimes fewer than 10, individuals—represents one of the most formidable challenges in modern medicine. For decades, the pharmaceutical industry has relied on the gold standard of randomized clinical trials (RCTs). However, when a patient population is so small that a traditional control group is statistically impossible or ethically untenable, the industry must pivot.
A paradigm shift is currently underway. Driven by the U.S. Food and Drug Administration’s (FDA) Rare Disease Evidence Principles (RDEP) process and the emerging "plausible mechanism framework," the regulatory landscape is evolving to prioritize flexibility without sacrificing scientific rigor.
Main Facts: The New Regulatory Reality
The core mission of modern rare disease drug development is to demonstrate "substantial evidence" of effectiveness. Historically, this has required at least two "adequate and well-controlled" investigations. For ultra-rare diseases, this requirement is being recalibrated.
The FDA’s current regulatory stance, bolstered by its 2023 guidance, acknowledges that a single pivotal trial, when supported by robust confirmatory evidence, can satisfy statutory standards. This is not an abandonment of standards, but a transition toward a risk-based, contextualized approach.
Key pillars of this new approach include:
- The Plausible Mechanism Framework: Where clinical benefit is observed in small cohorts, regulators are increasingly willing to accept evidence that links the drug’s molecular action to the biological cause of the disease.
- External Controls: In the absence of RCTs, researchers are utilizing natural history cohorts, high-quality registries, and observational real-world data (RWD) to construct synthetic control arms.
- Integrated Evidence Packages: Success now depends on the internal consistency of data across disparate sources—clinical trials, mechanistic studies, biomarkers, and patient-reported outcomes (PROs).
Chronology: The Evolution of Rare Disease Oversight
The movement toward these flexible frameworks did not happen overnight. It is the result of a multi-year effort to reconcile traditional FDA mandates with the harsh realities of orphan disease biology.
- Pre-2020: The industry struggled with the "small-n" problem, often leading to stalled development programs that could not meet the two-trial requirement.
- 2023: The FDA released pivotal guidance formally acknowledging that a single adequate trial, paired with confirmatory evidence, could serve as the basis for approval. This signaled a major departure from the "two-trial" orthodoxy for high-unmet-need areas.
- 2025–2026: The implementation of the RDEP process provided a structured mechanism for sponsors to engage with the FDA early in the development lifecycle. It formalized the dialogue process, ensuring that evidence packages are built with regulatory alignment from the outset.
- Current State: We are currently in an era where "fit-for-purpose" data strategies are being stress-tested in real-time, with regulators focusing on the quality of mechanistic evidence rather than the sheer volume of patients.
Supporting Data: Why "Small" Needs "Robust"
For ultra-rare conditions, the definition of "evidence" has expanded significantly. Because the patient sample size is limited, the depth of the data must increase.
The Role of Real-World Evidence (RWE)
RWE has become the backbone of the RDEP review process. It serves two primary functions:
- Establishing Prevalence: RWE helps prove that a disease meets the "ultra-rare" threshold, particularly for conditions that are frequently misdiagnosed or poorly coded in electronic health records (EHRs).
- Contextualizing Treatment: When a sponsor conducts a single-arm study, the "control" is often non-existent. RWE—derived from natural history studies—provides the necessary baseline to understand what would have happened to the patient in the absence of treatment.
Mechanistic Validation
As noted by experts like Julien Heidt of IQVIA, the goal is to create an evidentiary package that is internally consistent. If a patient shows clinical improvement, that improvement must be validated by secondary endpoints: pharmacodynamic markers, biomarker normalization, and non-clinical models that confirm the drug is hitting its intended molecular target.
Official Responses and Strategic Guidance
Regulatory bodies, including the FDA, have emphasized that RDEP is not an "easier" path, but a "different" one. It does not replace standard meeting milestones like end-of-Phase II or pre-NDA discussions; rather, it supplements them by providing a forum for early evidentiary alignment.
The Sponsor’s Checklist for Success
- Early Engagement: Before launching a pivotal trial, sponsors must submit an RDEP request under an existing Investigational New Drug (IND) application. This prevents the "evidentiary mismatch" where a company spends years on a study that the FDA deems insufficient.
- Standardization: Using shared clinical ontologies is critical. In a globalized trial, disparate coding systems can ruin data integrity. Standardized data models ensure that an outcome in a European clinic is interpreted the same way as an outcome in an American hospital.
- Privacy Governance: With datasets as small as ten patients, the risk of re-identification is extreme. Robust data governance must be baked into the trial design to maintain patient trust and legal compliance.
Implications: The Future of Drug Development
The shift toward the plausible mechanism framework and RDEP has profound implications for the biotechnology sector.
A Faster Path to Market?
While these mechanisms do not explicitly "expedite" timelines, they effectively shorten the path to approval by reducing the likelihood of a Complete Response Letter (CRL) caused by inadequate study design. By aligning on evidentiary requirements early, companies avoid the "re-do" cycle that has historically plagued orphan drug development.
Payer and Post-Market Realities
A major implication is the widening gap between regulatory approval and market access. Regulators may accept a single trial with mechanistic evidence, but payers (insurers and government health systems) often demand more, such as long-term durability data or quality-of-life metrics.
Sponsors must now think about the "total evidence plan." This includes:
- Patient-Reported Outcomes (PROs): Capturing the "lived experience" of the patient is now as important as the clinical readout.
- Clinician Education: Because ultra-rare diseases are often obscure, companies must pair their evidence generation with robust disease awareness campaigns.
- Post-Market Surveillance: Because the pre-approval dataset is smaller, the post-market phase becomes an essential period for collecting additional evidence to support long-term efficacy and safety.
The Human Element
Ultimately, these regulatory changes are about people. In the context of ultra-rare diseases, the patient is not just a data point; they are an active partner in the drug development process. By integrating patient advocacy groups into the early stages of trial design, sponsors gain invaluable insights into which endpoints actually matter to those living with the condition.
Conclusion: A Complex but Evolving Path
The development of therapies for ultra-rare diseases remains one of the most intellectually and logistically demanding fields in science. However, the maturation of the RDEP process and the widespread acceptance of the plausible mechanism framework offer a beacon of hope.
Success in this arena is no longer defined by the ability to conduct massive, multi-center RCTs. Instead, it is defined by the ability to tell a compelling, scientifically rigorous, and internally consistent story. By leveraging high-quality RWE, engaging regulators with transparency, and grounding clinical outcomes in the lived reality of patients, the pharmaceutical industry can continue to push the boundaries of what is possible—bringing life-changing therapies to populations that were once considered "too small to treat."
References
- FDA, "Rare Disease Evidence Principles," 2025/2026 guidelines.
- FDA, "Plausible Mechanism Framework for Individualized Therapies," Draft Guidance.
- Federal Food, Drug, and Cosmetic Act, Section 505(d).
- FDA, "Providing Clinical Evidence of Effectiveness for Human Drug and Biological Products," 2023.
- Natural History Studies for Drug Development: Guidance for Industry.
- FDA Approval Documentation for Onasemnogene abeparvovec-brve (Zolgensma).