A Breakthrough in Cardiovascular Research
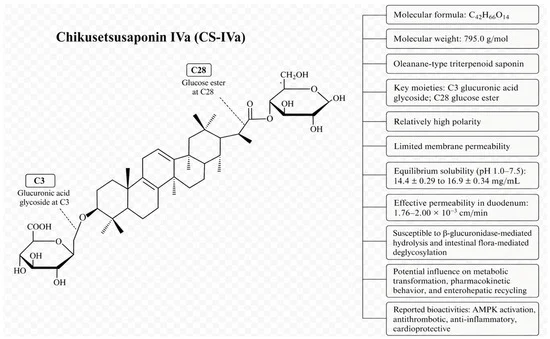
A recent study published in the journal Pharmaceuticals (Vol. 19, No. 5) has unveiled critical insights into the behavior of Chikusetsusaponin IVa (CS-IVa), a natural triterpenoid saponin, in the context of myocardial ischemia/reperfusion injury (MIRI). While CS-IVa has long been recognized for its cardioprotective potential, this research marks a significant departure from traditional efficacy studies by providing a rigorous, data-driven analysis of how the drug’s pharmacokinetic (PK) profile changes under pathological conditions.
The study, conducted by a team of researchers from the Guangzhou University of Chinese Medicine, addresses the "translational gap" that often plagues cardioprotective research. By combining conventional pharmacodynamic (PD) endpoints with an exploratory exposure–response framework, the authors have laid a more robust foundation for the potential clinical development of CS-IVa.

Chronology of the Investigation
The research followed a systematic methodology designed to move beyond basic efficacy and into the realm of mechanistic understanding:
- Model Establishment (March 2026): Researchers established a rat model of MIRI by ligating the left anterior descending coronary artery for 30 minutes, followed by a 24-hour reperfusion period.
- Pharmacokinetic Profiling (Spring 2026): Using UHPLC-MS/MS, the team quantified CS-IVa plasma concentrations over a 24-hour period, comparing systemic exposure in healthy rats versus those suffering from MIRI.
- Model Re-evaluation (May 2026): To ensure accuracy, the team conducted a systematic re-evaluation of candidate PK models, applying stringent criteria for stability and parsimony, ultimately selecting a one-compartment model.
- Publication (May 11, 2026): The study was officially published, offering a detailed analysis of how pathological states fundamentally alter drug disposition.
Supporting Data and Methodology
The study’s findings were built on a multi-faceted evaluation of cardiac health. Cardiac function was measured via echocardiography and hemodynamic parameters (+dp/dtmax and -dp/dtmax). The results were compelling: CS-IVa significantly improved left ventricular function and reduced the size of myocardial infarctions.

Pharmacokinetic Discrepancies
One of the most striking findings was the marked difference in drug behavior between healthy and diseased subjects. At a dose of 15 mg/kg, MIRI rats displayed significantly lower maximum plasma concentration (Cmax) and area under the curve (AUC) compared to healthy rats. Furthermore, the MIRI rats exhibited a delayed time to reach maximum concentration (Tmax) and a shortened apparent terminal half-life. These findings suggest that the metabolic and physiological stress induced by heart injury fundamentally changes how the body handles the compound.
The Exposure–Response Gap
The team observed a "counterclockwise hysteresis" pattern when mapping plasma concentration against CK-MB—a biomarker for heart muscle injury. This indicated that the therapeutic effect (the reduction of CK-MB levels) did not occur in real-time with plasma drug levels, suggesting a time-dependent delay in the drug’s mechanism of action. By utilizing an effect-compartment approach, the researchers determined that the drug requires a period of distribution or biological transduction before its full cardioprotective effect is realized.

Scientific Implications and Limitations
The study is notable for its candid approach to modeling. The researchers explicitly categorized their exposure–response findings as "exploratory" rather than "mechanistic," acknowledging that biomarkers like CK-MB are secondary indicators of injury rather than direct drug targets.
Key Implications:

- Disease-State Awareness: The research underscores that pharmacokinetics established in healthy models cannot be safely extrapolated to clinical patients suffering from acute ischemia.
- Model Stability: The use of "leave-one-rat-out" robustness analysis provides a template for future preclinical studies to ensure their PK models are not skewed by individual animal outliers.
- Data-Driven Future: While the team utilized artificial intelligence models (such as Graph Attention Networks) as supplementary tools, they cautioned that these are not yet ready to replace mechanistic biological models, particularly given the small sample sizes typical in preclinical settings.
Official Perspectives and Future Directions
The research team, led by Dr. Lijun Zhu and Dr. Yuanyuan Cheng, emphasizes that while the findings are promising, they are a starting point. "The translational relevance of our findings requires cautious interpretation," the authors noted in their discussion. They highlighted that the rat model, while essential for controlled observation, does not replicate the complexity of human patients—who often face comorbidities, polypharmacy, and variable genetic backgrounds.
Future Requirements:

- Clinical Formulation: Future studies must transition to human-relevant delivery methods, as the current study utilized intraperitoneal injection.
- Individual-Level Data: Researchers emphasize the need for continuous, individual-level monitoring of PD endpoints to refine the current group-level models.
- Tissue Distribution: Further investigation into how CS-IVa is distributed into myocardial tissue—as opposed to just plasma—is required to bridge the gap between systemic exposure and actual cardiac protection.
In conclusion, this study provides a vital reality check for the field of cardioprotective pharmacology. By proving that the disease state itself changes the drug’s "journey" through the body, the researchers have set a new standard for how experimental drugs should be evaluated before they reach the clinic. As the scientific community continues to seek better treatments for myocardial infarction, such rigorous, transparency-focused research will be essential to ensure that promising compounds like Chikusetsusaponin IVa can eventually offer real-world benefits to patients.