By Investigative Health Reporter
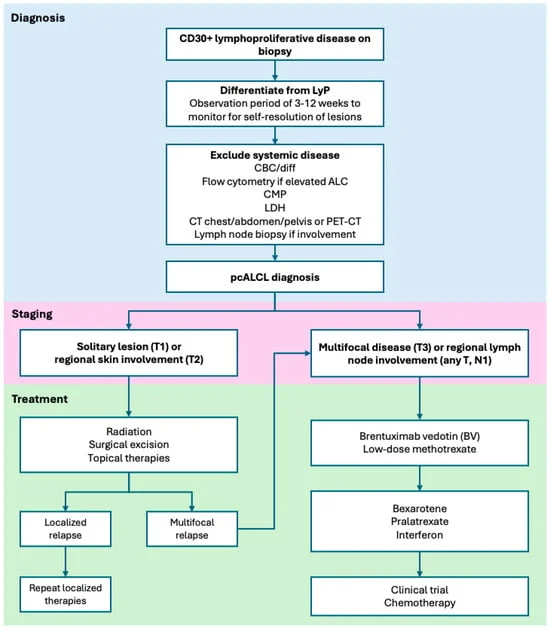
The landscape of oncology has undergone a radical transformation over the last decade, shifting from a "one-size-fits-all" approach to a highly nuanced era of precision medicine. For patients diagnosed with early-stage breast cancer, the influx of information can be as daunting as the diagnosis itself. Central to this modern approach are two distinct yet frequently conflated diagnostic tools: genetic testing and biomarker testing.
While the terms are often used interchangeably in casual conversation, they represent two different pillars of cancer care. One looks backward at a patient’s ancestry and inherited risk, while the other looks forward at the specific behavior of a tumor. Understanding the divergence between these two is no longer just a matter of medical semantics; it is a critical component of tailoring a treatment plan that maximizes efficacy while minimizing unnecessary toxicity.
Main Facts: Defining the Two Pillars of Precision Oncology
To navigate a breast cancer diagnosis, one must first understand the biological "location" of the data being analyzed.
Genetic Testing: The Hereditary Map
Genetic testing, often referred to as germline testing, examines the DNA a person was born with—the genetic code present in every cell of their body. This DNA is inherited from biological parents and can be passed down to children. In the context of breast cancer, genetic testing looks for specific mutations, such as BRCA1, BRCA2, PALB2, or CHEK2, which significantly increase the lifetime risk of developing breast and ovarian cancers.
The primary goal of genetic testing is to identify a hereditary predisposition. This information is foundational for long-term health management, influencing decisions about prophylactic (preventative) surgeries, the frequency of screenings, and the health surveillance of family members who may share the same genetic markers.
Biomarker Testing: The Tumor’s Signature
Biomarker testing, also known as genomic or somatic testing, focuses exclusively on the DNA and RNA within the cancerous tumor itself. Unlike the DNA you are born with, these genetic changes are "acquired" or "somatic," meaning they exist only within the malignant cells.
Tests such as Agendia’s MammaPrint and BluePrint analyze the expression of specific genes within the tumor to determine its "personality." This testing answers the most pressing questions following a diagnosis: How aggressive is this specific cancer? Is it likely to return after surgery? Will this specific tumor respond to chemotherapy, or is the patient better served by hormone therapy alone?
Chronology: The Diagnostic Journey
The integration of these tests follows a specific timeline in the standard of care for early-stage breast cancer.
- Detection and Biopsy: The journey typically begins with a screening mammogram or the discovery of a lump, followed by a core needle biopsy to confirm malignancy.
- The Genetic Crossroads: At the point of diagnosis, a physician evaluates the patient’s family history and age. If there is a strong history of cancer or if the patient is particularly young, genetic testing is often ordered immediately. This helps determine if the patient should undergo a lumpectomy or a more extensive bilateral mastectomy to prevent future occurrences.
- Surgical Intervention: The primary tumor is removed. Following surgery, the pathology report provides the first glimpse into the cancer’s characteristics (Stage, Grade, and Hormone Receptor status).
- The Genomic Window (Biomarker Testing): Once the tumor is removed, a sample is sent for biomarker testing. This occurs in the "decision window"—the few weeks between surgery and the start of systemic treatment (chemotherapy or radiation).
- The Treatment Roadmap: Using the results from the biomarker tests, the oncology team and the patient finalize the treatment plan. This ensures that chemotherapy is reserved only for those whose tumor biology necessitates it.
Supporting Data: The Shift Toward De-escalation
The push for biomarker testing is backed by robust clinical data that suggests many women with early-stage breast cancer have been historically over-treated. For decades, chemotherapy was the standard "safety net" for almost all breast cancer patients. However, large-scale clinical trials have changed this paradigm.
One of the most significant studies in this field is the MINDACT trial, a randomized Phase III controlled trial. The study utilized the MammaPrint 70-gene signature to evaluate over 6,000 women. The data revealed that a significant percentage of women who were clinically "high risk" (based on tumor size or lymph node involvement) were actually "genomically low risk."
The findings were profound: women with a "Low Risk" MammaPrint result had excellent outcomes—with a 5-year distant metastasis-free survival rate of over 94%—regardless of whether or not they received chemotherapy. This data provides the scientific "permission" for oncologists to safely de-escalate treatment, sparing thousands of women the grueling side effects of chemotherapy, such as hair loss, nausea, neuropathy, and long-term heart damage.
Furthermore, Agendia’s BluePrint test provides a second layer of data by identifying the "functional subtype" of the cancer. While traditional pathology might label a tumor as "Luminal" based on protein stains, BluePrint looks at the underlying molecular activity, occasionally revealing that a tumor is behaving more like a "Basal-type" or "HER2-type," which requires a completely different therapeutic approach.
Official Responses and Expert Perspectives
Organizations like Sharsheret, a national non-profit supporting Jewish women and families facing breast and ovarian cancer, emphasize the importance of education in this space. Because certain populations—such as those of Ashkenazi Jewish descent—have a higher prevalence of BRCA mutations (1 in 40 compared to 1 in 400 in the general population), Sharsheret advocates for early genetic testing.
"Understanding your genetic risk is about the ‘why’ and the ‘who else’ in your family," a spokesperson for the organization noted. "But once the diagnosis is on the table, biomarker testing becomes the ‘how’—how do we treat this specific tumor effectively?"
Medical device and diagnostic companies like Agendia have positioned their platforms not just as tests, but as "decision-support tools." By focusing on early-stage (Stage I and II) invasive breast cancer, these companies aim to provide clarity when the path forward is ambiguous.
Leading oncologists argue that these tests are now essential. According to current NCCN (National Comprehensive Cancer Network) guidelines, gene expression profiling is recommended for many patients with hormone-receptor-positive, HER2-negative early-stage breast cancer. The consensus among the medical community is shifting: failing to utilize biomarker testing may result in suboptimal care, either through over-treatment or under-treatment.
Implications: The Future of Personalized Care
The implications of distinguishing between genetic and biomarker testing extend beyond the individual patient to the broader healthcare system.
1. Financial Toxicity
Chemotherapy is expensive, both in terms of drug costs and the management of its side effects. By using biomarker testing to identify patients who do not benefit from chemotherapy, the healthcare system can save billions of dollars while improving patient quality of life.
2. Psychological Well-being
The "fear of recurrence" is a dominant psychological burden for survivors. Genetic testing provides clarity on the risk of new cancers, while biomarker testing provides a data-driven percentage of the risk of the original cancer returning. For many, a "Low Risk" genomic score provides a level of peace that clinical statistics alone cannot offer.
3. Precision in Surgery vs. Systemic Therapy
If a patient discovers they have a BRCA mutation (genetic testing), they might opt for a double mastectomy. However, that surgery does not dictate whether they need chemotherapy. Only the tumor’s biology (biomarker testing) can answer that. The implication is clear: a patient could potentially have an aggressive surgery but a "gentle" systemic treatment plan, or vice versa, depending on the results of both tests.
Conclusion: Smarter, Not Just Stronger
The mantra of modern oncology is to be "smarter, not just stronger." In the past, "stronger" meant the most aggressive surgery and the most toxic drugs. Today, "smarter" means using the dual lenses of genetic and biomarker testing to see the full picture.
Genetic testing allows patients to understand their heritage and protect their future and their families. Biomarker testing allows patients to understand their present—the specific enemy they are fighting—and choose the most effective weapon.
As we move further into the 2020s, the integration of these tests will likely become even more sophisticated. But for the patient standing at the threshold of a new diagnosis today, the message is simple: Ask your doctor for both. Know your inherited risk, and know your tumor’s personality. In the intersection of those two data points lies the path to a personalized, effective, and empowered recovery.