A groundbreaking study published in the journal Cancers has unveiled the promising role of atovaquone, an FDA-approved drug, in enhancing the effectiveness of radiation therapy for Diffuse Intrinsic Pontine Glioma (DIPG), a devastating pediatric brain tumor. Researchers have demonstrated that atovaquone, by targeting mitochondrial energy production, can sensitize DIPG cells to radiation, offering a potential new avenue for treatment. The study also introduces an advanced formulation of atovaquone designed for improved brain penetration, which showed significant efficacy in preclinical models.
The Critical Need for New DIPG Treatments
Diffuse Intrinsic Pontine Glioma (DIPG) represents a particularly aggressive form of pediatric brain cancer, notorious for its grim prognosis and limited treatment options. Despite advancements in medicine, DIPG remains largely untreatable, with a median survival rate of less than a year following diagnosis. Radiotherapy is currently the standard of care, offering temporary relief by slowing tumor growth, but its effectiveness is significantly hampered by the inherent radioresistance of DIPG cells. A key factor contributing to this resistance is believed to be the presence of low oxygen levels, or hypoxia, within the tumor microenvironment. This hypoxia shields tumor cells from the damaging effects of radiation, allowing them to survive and proliferate.

Atovaquone: A Novel Approach to Overcoming Radioresistance
The research team, led by Dr. Faiqa Mudassar and Dr. Kristina M. Cook from the Translational Radiation Biology and Oncology Laboratory at The Westmead Institute for Medical Research, focused on atovaquone, a drug already approved by the U.S. Food and Drug Administration (FDA) for treating malaria and Pneumocystis jirovecii pneumonia. Atovaquone’s known mechanism of action involves inhibiting mitochondrial oxidative phosphorylation (OXPHOS), a critical process for cellular energy production. By blocking OXPHOS, atovaquone reduces cellular oxygen consumption, potentially alleviating hypoxia and thereby increasing tumor sensitivity to radiation.
"Our work builds upon the understanding that DIPG tumors often exhibit characteristics of oxidative metabolism and hypoxia, both of which contribute to their resistance to standard treatments like radiation," explained Dr. Mudassar. "By targeting the very machinery that fuels these tumor cells, we hypothesized that atovaquone could create a more favorable environment for radiation therapy to succeed."

Unveiling the Mechanism: From Lab Bench to Preclinical Models
The study employed a multi-faceted approach, utilizing patient-derived DIPG cell lines and sophisticated laboratory techniques.
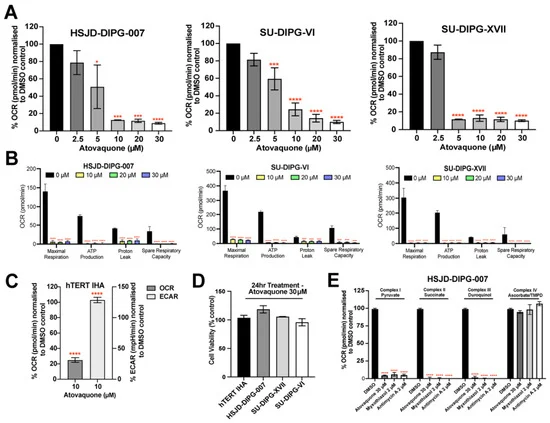
- Mitochondrial Respiration Inhibition: Extracellular flux analysis confirmed that atovaquone effectively suppressed mitochondrial respiration in DIPG cells, significantly reducing oxygen consumption rates. This effect was observed at concentrations that did not immediately induce cell death, suggesting a targeted metabolic intervention.
- Hypoxia Reduction: In three-dimensional neurosphere models, which better mimic the tumor microenvironment, atovaquone treatment led to a notable decrease in hypoxia markers. This was further supported by a reduction in the expression of HIF-1α, a key protein activated under low oxygen conditions and known to promote radioresistance.
- Enhanced Radiosensitivity: In vitro clonogenic survival assays demonstrated that atovaquone significantly enhanced the radiosensitivity of DIPG cells. When combined with radiation, atovaquone treatment resulted in a greater reduction in tumor cell survival compared to either treatment alone.
- Oxidative Stress and Transcriptomic Changes: Beyond hypoxia modulation, atovaquone also induced oxidative stress in DIPG cells, particularly at higher concentrations. Furthermore, transcriptomic and metabolomic analyses revealed broad molecular reprogramming, including the disruption of metabolic pathways, the activation of stress responses, and the suppression of cell cycle and DNA replication programs, especially when combined with radiation.
Overcoming the Blood-Brain Barrier Challenge
A significant hurdle for many potential brain tumor therapies is their ability to cross the blood-brain barrier (BBB) and reach therapeutic concentrations within the central nervous system. Recognizing this challenge, the researchers investigated an amorphous solid dispersion (ASD) formulation of atovaquone. This advanced formulation is designed to improve the drug’s solubility and bioavailability, thereby enhancing its penetration into the brain.

"We understood that for atovaquone to be truly effective against brain tumors like DIPG, we needed to ensure it could reach the tumor site in sufficient quantities," stated Dr. Cook. "The ASD formulation represents a significant step forward in addressing this pharmacokinetic challenge."
The ASD atovaquone formulation demonstrated comparable in vitro efficacy to the standard formulation. Crucially, pharmacokinetic studies in mice revealed that the ASD formulation achieved measurable concentrations of atovaquone in the brain, including the brainstem, where DIPG tumors are located.

Promising Preclinical Efficacy in an Orthotopic Model
To assess the therapeutic potential of the ASD atovaquone formulation in a more complex biological setting, the researchers utilized an orthotopic DIPG mouse model. In this model, human DIPG cells were implanted directly into the brainstem of mice. The results were encouraging: while neither radiation nor ASD atovaquone alone significantly extended survival, the combination of ASD atovaquone with radiation therapy led to a statistically significant prolongation of survival. This finding provides critical preclinical validation for atovaquone as a radiosensitizing agent for DIPG and highlights the importance of advanced drug formulations for CNS-targeted therapies.
Official Statements and Expert Opinions
"This research represents a vital step forward in our understanding of how to combat DIPG," commented Dr. Eric Hau, a senior author on the study. "The fact that atovaquone, an existing and well-tolerated drug, shows such promise in combination with radiation is incredibly encouraging. The development of an improved formulation for brain penetration further strengthens its potential for clinical translation."

The study’s funding sources, including the NSW Health, Cancer Institute NSW, and various pediatric cancer foundations, underscore the significant collaborative effort and investment in finding solutions for DIPG. Organizations like The Cure Starts Now Foundation and the DIPG/DMG Collaborative have been instrumental in supporting research that pushes the boundaries of treatment.
Implications for Future DIPG Treatment
The findings of this study have profound implications for the future management of DIPG and potentially other pediatric brain tumors. By targeting mitochondrial metabolism, researchers are opening up a new therapeutic paradigm that complements existing treatments like radiation. The demonstrated ability of the ASD formulation to improve brain exposure is a critical advancement that could translate into more effective treatments for brain cancers.

The researchers propose that atovaquone could be considered as a radiosensitizing agent in future clinical trials for DIPG. Further research will focus on optimizing dosing strategies, refining delivery methods, and identifying biomarkers that can predict patient response to atovaquone-based therapies. The established safety profile of atovaquone in both children and adults is a significant advantage, potentially allowing for earlier and broader clinical investigation.
While the modest survival benefit observed in the mouse model warrants further optimization, the proof-of-concept is compelling. This research offers a beacon of hope for families affected by DIPG, suggesting that a combination therapy approach, leveraging the power of an existing drug in a novel way, could one day lead to improved outcomes for these young patients. The scientific community eagerly anticipates the progression of this promising research towards clinical trials.