Glioblastoma (GBM), the most lethal primary brain malignancy, has long frustrated oncologists and researchers. Characterized by aggressive invasiveness and a profound resistance to standard therapies, GBM creates an immunosuppressive environment that effectively neutralizes the body’s natural defenses. A groundbreaking study recently published in the journal Cancers (May 2026) offers a new perspective on why immunotherapy often fails in these patients. By modeling chronic exposure to interferon-gamma (IFN-γ)—the immune system’s primary “alert” signal—researchers have discovered that tumor cells do not simply die; they adapt, rewire, and enter distinct states of persistent survival.
The Paradox of Interferon-Gamma in Glioblastoma
In a healthy immune response, IFN-γ acts as a beacon, signaling T-cells to identify and eliminate malignant cells. However, in the chaotic environment of a glioblastoma tumor, this same signal can paradoxically become a tool for tumor preservation.

The research team, led by Dr. Santosh Kesari and Elnaz Rahbarlayegh of the Pacific Neuroscience Institute and The Lundquist Institute, sought to understand the long-term impact of this signaling. While acute exposure to IFN-γ typically suppresses tumor growth, chronic exposure creates a "conditioned" state. This state fosters resistance to therapy and maintains an immunosuppressive microenvironment, explaining why "turning up the volume" of the immune system via conventional immunotherapy has struggled to provide durable clinical benefits for GBM patients.
Chronology of Adaptation: A 28-Day Study
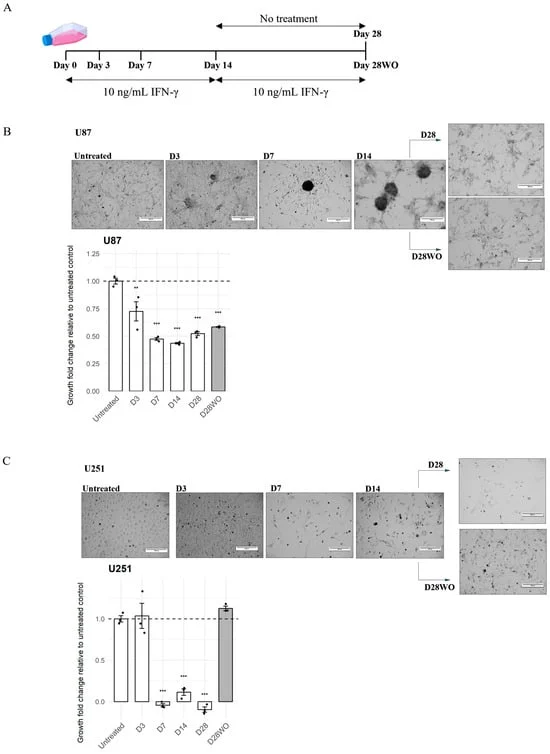
To capture the evolution of this resistance, the researchers conducted a 28-day longitudinal study using two distinct GBM cell lines: the mesenchymal-like U87 line and the proneural-like U251 line.

- Initial Phase (Days 0–3): Both cell lines responded to IFN-γ with a canonical immune activation. Antigen presentation pathways were upregulated, and cell growth was initially suppressed.
- Intermediate Phase (Day 14): As exposure continued, the cell lines began to diverge. U87 cells showed a partial, non-linear attenuation of the immune response, while U251 cells began to display a more rigid, sustained interferon-driven state.
- Chronic Phase (Days 14–28): The divergence became pronounced. U87 cells adopted a "persistence-prone" state, characterized by an unexpected activation of the PI3K-AKT signaling pathway—a key survival and growth mechanism. Conversely, U251 cells leaned into an "Interferon-Related DNA-Damage Resistance Signature" (IRDS), which made them more resilient to DNA damage and therapy.
- The Washout Period: When the cytokine was removed, the cells did not return to their baseline. The "interferon-imprinting" was stable; the cells had been fundamentally reprogrammed, showing that the adaptive changes were not merely temporary responses to an external stimulus but rather a long-term alteration of the tumor cell identity.
Supporting Data: Signaling and Secretome Shifts
The strength of the study lies in its multi-omic approach, integrating transcriptomic, proteomic, and secretome profiling.
Automated Western blotting and single-cell proteomics revealed that while IFN-γ increases STAT1 phosphorylation in both lines, the "wiring" of the cell changes differently. In U87 cells, the signaling network tightened, with cells increasingly co-activating multiple survival proteins simultaneously. In U251 cells, the shift was more selective, focusing on an IRDS-driven inflammatory state.

Furthermore, the "secretome"—the cocktail of signaling proteins the tumor releases into its microenvironment—showed that U87 cells evolved to output a broad, pro-inflammatory mix. In contrast, U251 cells adopted a more targeted, CCL2-dominant profile. CCL2 is a notorious protein in cancer biology known for recruiting suppressive myeloid cells into the tumor, effectively creating a "shield" that prevents T-cells from performing their duties.
Clinical Relevance: From Lab to Patient
The researchers validated these findings by analyzing the Cancer Genome Atlas (TCGA) dataset of 172 glioblastoma patients. The data confirmed that the adaptive programs identified in the petri dish are present in human tumors.

The team found that high interferon signaling in human GBM patients correlates strongly with PI3K-AKT pathway activity. Even more concerning, tumors that exhibited this specific survival-signaling signature were associated with reduced overall survival. Furthermore, these interferon-active tumors were consistently enriched with signatures of T-cell exhaustion and macrophage infiltration, providing a clear map of why these tumors remain "cold" or immunosuppressive despite the presence of immune activity.
Implications for Future Therapeutic Strategies
The findings present a paradigm shift in how we might treat glioblastoma. The researchers suggest that the "one-size-fits-all" approach to immunotherapy is likely insufficient because the tumor’s response is highly dependent on its specific molecular lineage.

- Context-Dependent Combination Therapy: For tumors with a CCL2-dominant, IRDS-persistent profile (like the U251 model), the authors suggest that therapies targeting the CCR2/CCL2 axis might be necessary to break the immunosuppressive shield.
- Targeting Survival Pathways: For tumors that, like the U87 model, default to PI3K-AKT survival signaling under immune pressure, combining immunotherapy with PI3K/AKT inhibitors could prevent the tumor from "adapting away" the benefits of the immune response.
- Temporal Precision: The study highlights that the duration of immune priming matters. The authors propose that time-bounded, strategic exposure to interferon might capture the benefits of antigen presentation without allowing the tumor enough time to fully establish its adaptive, therapy-resistant state.
As oncology moves toward a more personalized approach, this research underscores a critical realization: the tumor is not a static target. It is a dynamic, evolving entity that learns to survive under the pressure of our best treatments. By decoding the specific "adaptive language" of glioblastoma subtypes, clinicians may soon be able to outmaneuver these defenses, turning the immune system’s signal from a paradoxical shield into a weapon of destruction.
Summary of Key Findings
- Lineage-Dependent Adaptation: Glioblastoma cell lines adapt to chronic immune signaling differently based on their molecular state (Mesenchymal vs. Proneural).
- Incomplete Reversibility: Once tumor cells are "conditioned" by interferon, they do not easily revert, even after the signal is removed.
- Survival Crosstalk: Interferon signaling is fundamentally linked to the PI3K-AKT survival pathway in human GBM, explaining why these tumors survive immune-mediated stress.
- Clinical Potential: The study provides a rationale for combination therapies that simultaneously target the immune pathway and the specific survival-rewiring mechanism activated by the tumor.