Introduction
Esophageal squamous cell carcinoma (ESCC) stands as a formidable global health challenge, characterized by its aggressive nature and limited therapeutic options. The urgent need for reliable experimental models to unravel its complexities and accelerate the development of effective treatments is paramount. A recent study published in the journal Cancers sheds crucial light on the establishment and characterization of a preclinical ESCC xenograft model, offering practical guidance for researchers seeking to enhance the reproducibility and translational relevance of their findings. The research meticulously investigates how different implantation conditions impact tumor growth, histological structure, and cellular viability, ultimately providing a more robust platform for preclinical cancer studies.

Main Facts
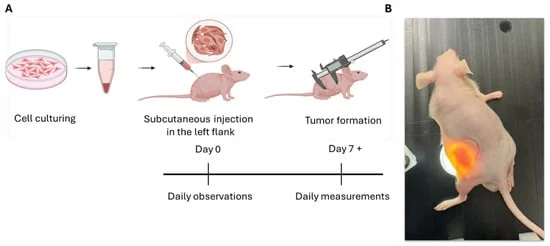
A team of researchers, led by Pavel A. Solopov, has detailed the establishment and characterization of a subcutaneous xenograft model using the human esophageal squamous cell carcinoma (ESCC) cell line KYSE-30 in immunodeficient J:NU mice. The study systematically compared three distinct implantation conditions: cells suspended in phosphate-buffered saline (PBS), Matrigel (a basement membrane matrix), and a combination of Matrigel and PBS.

The key findings reveal that the choice of implantation matrix significantly influences tumor development. While Matrigel alone led to rapid tumor growth, it limited the practical window for therapeutic interventions. Conversely, PBS alone resulted in inefficient tumor engraftment and increased necrosis. The most promising approach identified was the use of Matrigel diluted with PBS, which facilitated consistent tumor establishment with a balanced and reproducible growth rate, ideal for preclinical studies.
Histopathological analysis further elucidated these differences, showing that matrix-supported tumors exhibited reduced necrosis, more organized stromal architecture, and enhanced proliferative activity compared to tumors grown in PBS. These microenvironmental factors are critical for understanding tumor biology and predicting treatment responses. The study underscores the importance of meticulous histological evaluation to ensure the reliability of preclinical cancer research.

Chronology of the Study
The research project involved a series of well-defined stages to ensure a comprehensive understanding of the xenograft model’s behavior:

- Cell Culture and Preparation: Human ESCC KYSE-30 cells were cultured under standard laboratory conditions. On the day of implantation, cells were harvested, counted, and prepared for injection.
- Preparation for Implantation: Cells were suspended in one of three formulations: PBS, Matrigel, or a 1:1 mixture of Matrigel and PBS. These suspensions were kept on ice to maintain cell viability.
- Animal Studies and Implantation: Immunodeficient J:NU mice were housed and acclimated. Following ethical approval, mice were manually restrained, and the left flank was disinfected. A precisely measured volume of cell suspension (1.5 x 106 cells in 50 µL) was injected subcutaneously into the flank using a fine-gauge needle. A control group received Matrigel without cells.
- Tumor Monitoring: Beginning on day 5 post-implantation, tumors were visually inspected daily. Tumor size was measured using digital calipers, recording the longest and shortest perpendicular diameters to calculate tumor volume.
- Study Endpoint and Euthanasia: Mice were monitored until predefined experimental endpoints were met, typically a target tumor volume of 50-100 mm3 for therapeutic intervention studies, or a maximum of 400 mm3. Humane endpoints were also strictly observed. Euthanasia was performed ethically using isoflurane inhalation followed by cervical dislocation.
- Tissue Collection and Processing: Upon necropsy, tumors were excised, photographed, and then fixed in 10% neutral-buffered formalin for 72 hours. The fixed tissues were subsequently embedded in paraffin, sectioned at 5 µm thickness, and mounted on glass slides for analysis.
- Histological and Immunohistochemical Analyses: Serial sections were subjected to Hematoxylin and Eosin (H&E) staining for general morphology, Masson’s trichrome staining for stromal collagen deposition, Pan-cytokeratin (Pan-CK) for epithelial confirmation, Ki-67 for proliferative activity, CD31 for microvascular density, and TUNEL assay for apoptotic index.
- Data Analysis: Histopathological and immunohistochemical quantifications were performed on viable tumor regions, excluding necrotic areas, using Fiji (ImageJ) software. Statistical analyses were conducted to compare findings across different experimental groups.
Supporting Data: A Deeper Dive into the Microenvironment
The study’s rigorous methodology yielded significant data that illuminate the impact of implantation matrices on tumor biology:

- Tumor Growth Dynamics: The Matrigel + PBS group demonstrated a controlled growth pattern, reaching the target experimental volume within 7-14 days, a crucial aspect for therapeutic studies. In contrast, Matrigel alone resulted in very rapid growth, potentially outgrowing the experimental window, while PBS alone showed poor tumor establishment.
- Necrosis and Viability: H&E staining revealed substantial central necrosis in tumors grown in PBS, indicative of poor vascularization and nutrient supply. Matrigel-supported tumors, however, exhibited significantly reduced necrotic fractions, suggesting better structural integrity and viability.
- Stromal Architecture and Collagen Deposition: Masson’s trichrome staining highlighted differences in the extracellular matrix. Tumors in Matrigel displayed more organized collagen networks, indicating matrix-driven stromal remodeling, which can influence tumor mechanics and therapeutic response.
- Epithelial Identity: Pan-cytokeratin staining confirmed that the epithelial identity of the KYSE-30 cells remained consistent across all implantation conditions, assuring the integrity of the tumor model.
- Vascularization: CD31 immunostaining indicated that Matrigel-supported tumors had more developed and evenly distributed microvasculature compared to the sparse vascularization observed in PBS-derived tumors. This is critical for nutrient and drug delivery.
- Proliferative Activity: Ki-67 staining revealed a significantly higher proliferation index in Matrigel-supported tumors, correlating with their faster and more robust growth.
- Apoptotic Activity: TUNEL assay showed only modest differences in baseline apoptosis among the groups, suggesting that proliferative activity, rather than apoptosis, was the primary driver of observed growth differences.
Official Responses and Implications
While the provided content does not include direct "official responses" from external bodies, the publication itself in Cancers, a reputable peer-reviewed journal, signifies endorsement and validation by the scientific community. The implications of this research are far-reaching for the field of esophageal cancer preclinical research:

- Enhanced Reproducibility: By clearly defining and recommending an optimal implantation strategy (Matrigel + PBS), the study provides a blueprint for researchers to establish more reproducible xenograft models. This consistency is vital for comparing results across different laboratories and for the reliable translation of findings from bench to bedside.
- Improved Model Relevance: The detailed histopathological characterization allows for a deeper understanding of how implantation conditions shape the tumor microenvironment. This knowledge is crucial for selecting appropriate models for specific research questions, particularly those involving therapies that are sensitive to tumor architecture, vascularization, or matrix composition.
- Guidance for Therapeutic Studies: The identification of an implantation method that balances reliable engraftment with a controlled growth window directly benefits researchers planning therapeutic intervention studies. This ensures that experiments can be conducted with sufficient time for treatment efficacy to manifest without tumors becoming excessively large or necrotic.
- Foundation for Future Research: This work serves as a foundational characterization for the KYSE-30 xenograft model. Future studies can build upon this established baseline to investigate novel therapeutic strategies, mechanisms of resistance, or the role of specific microenvironmental factors in ESCC progression.
Conclusion
The study by Solopov and colleagues represents a significant contribution to the advancement of esophageal cancer research. By systematically dissecting the impact of implantation conditions on a well-established ESCC xenograft model, they have provided invaluable insights into optimizing preclinical study designs. The recommendation of using Matrigel diluted with PBS offers a clear path towards more reproducible and biologically relevant xenograft models. This meticulous approach to model characterization is essential for accelerating the development of effective treatments for esophageal squamous cell carcinoma and ultimately improving patient outcomes. The detailed histological and immunohistochemical data presented offer a robust foundation for future investigations into the complex biology of this aggressive disease.