Editorial Note: This report was updated on May 11, 2026, to incorporate significant revisions to the Trump administration’s implementation timeline for the BALANCE Model and the extension of the Medicare GLP-1 Bridge program through 2027.
The rapid ascent of GLP-1 receptor agonists—a class of medications including semaglutide (Ozempic, Wegovy, Rybelsus) and tirzepatide (Mounjaro, Zepbound)—has reshaped the landscape of modern medicine. Once reserved for type 2 diabetes management, these drugs have demonstrated profound efficacy in treating obesity, cardiovascular disease, and sleep apnea. Yet, for millions of Americans, these clinical breakthroughs remain financially out of reach. According to recent KFF polling, 56% of GLP-1 users report significant difficulty affording their prescriptions, with one in four labeling the cost as "very difficult" to manage.
As the demand for these life-altering medications climbs, the Trump administration has embarked on a multi-pronged strategy to reduce costs and broaden access. However, recent developments indicate a pivot in policy, marked by the indefinite delay of the much-anticipated "Better Approaches to Lifestyle and Nutrition for Comprehensive hEalth" (BALANCE) Model in Medicare, coupled with a strategic extension of the temporary Medicare GLP-1 Bridge program.
The Landscape of Coverage: A Legal and Financial Quagmire
The central challenge in expanding GLP-1 access lies in the rigid statutory framework governing federal health programs. Since the inception of the Medicare Part D prescription drug benefit, federal law has explicitly prohibited the coverage of medications when used exclusively for weight loss. While Part D plans must cover two drugs per therapeutic category, obesity-related treatments remain categorically excluded. Beneficiaries can currently only access these drugs if they meet the criteria for a separate, FDA-approved indication, such as type 2 diabetes or cardiovascular risk reduction.

In contrast, the Medicaid landscape is defined by state-level discretion. Under the Medicaid Drug Rebate Program (MDRP), states must cover most FDA-approved drugs; however, federal law leaves the decision to cover weight-loss medication entirely to the states. As of January 2026, only 13 states provide such coverage—a decline from 16 states in 2025. This contraction is largely attributed to severe state budget constraints and the cascading effects of federal funding cuts, forcing state legislatures to prioritize immediate fiscal stability over the long-term health benefits of obesity management.
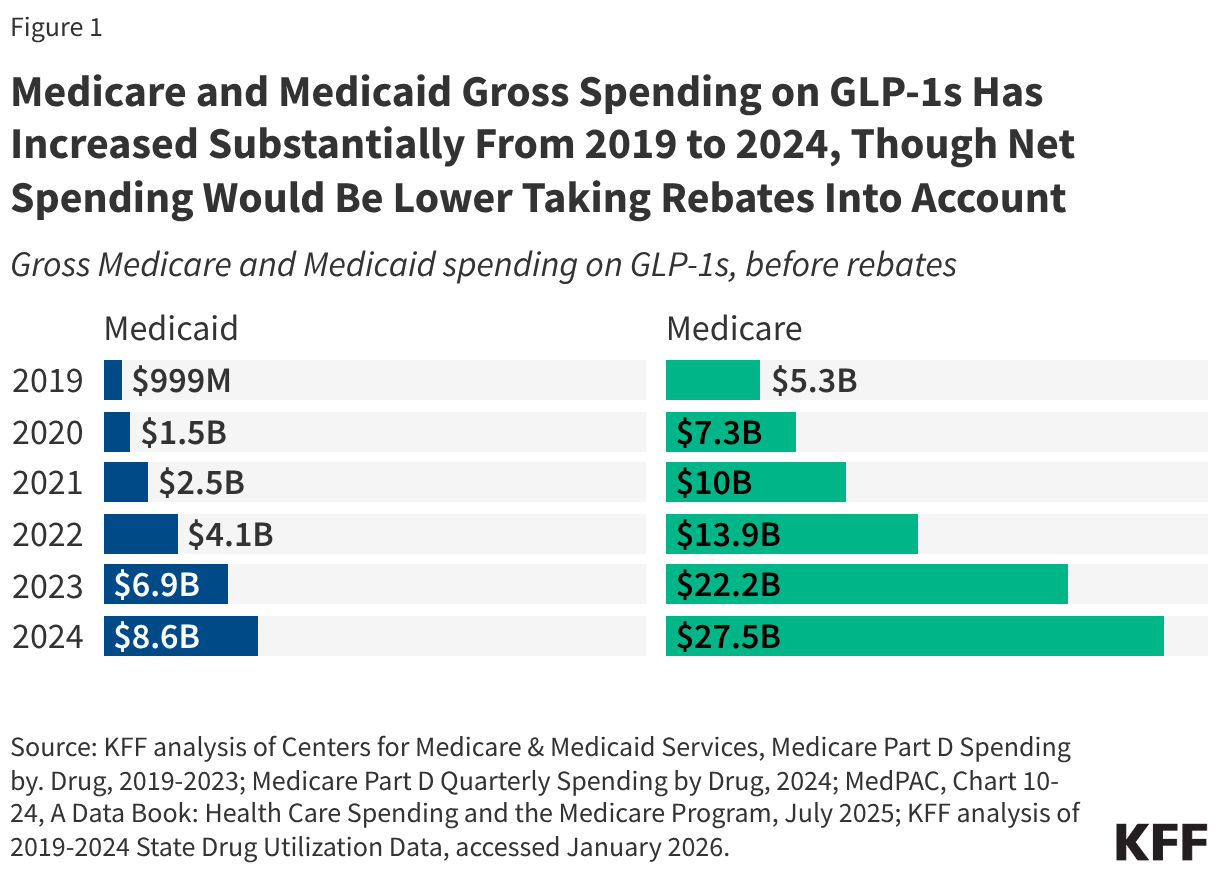
Despite these hurdles, spending on GLP-1s has surged. In 2024 alone, Medicaid recorded $8.6 billion in gross spending on these drugs, while Medicare saw an staggering $27.5 billion in gross spending across 21.8 million claims.
Chronology: From Ambition to Realignment
The administration’s policy trajectory has undergone several critical phases over the last 18 months:
- Late 2025: The White House announced "most-favored nation" pricing deals with pharmaceutical giants Novo Nordisk and Eli Lilly, aimed at curbing the retail costs of GLP-1s through initiatives like the TrumpRx platform.
- January 8, 2026: Deadline for manufacturers to signal interest in the BALANCE Model. Both Novo Nordisk and Eli Lilly agreed to participate.
- April 20, 2026: The deadline for Medicare Part D plan sponsors to apply for the BALANCE Model.
- April 21, 2026: In a major policy shift, CMS announced the indefinite delay of the BALANCE Model for Medicare Part D, citing insufficient interest from insurers, and simultaneously extended the GLP-1 Bridge program through the end of 2027.
- May 1, 2026: The BALANCE Model officially launches for state Medicaid programs, running through December 2031.
- July 31, 2026: Final deadline for state Medicaid agencies to submit their applications to participate in the BALANCE Model.
The Medicare GLP-1 Bridge: A Temporary Lifeboat
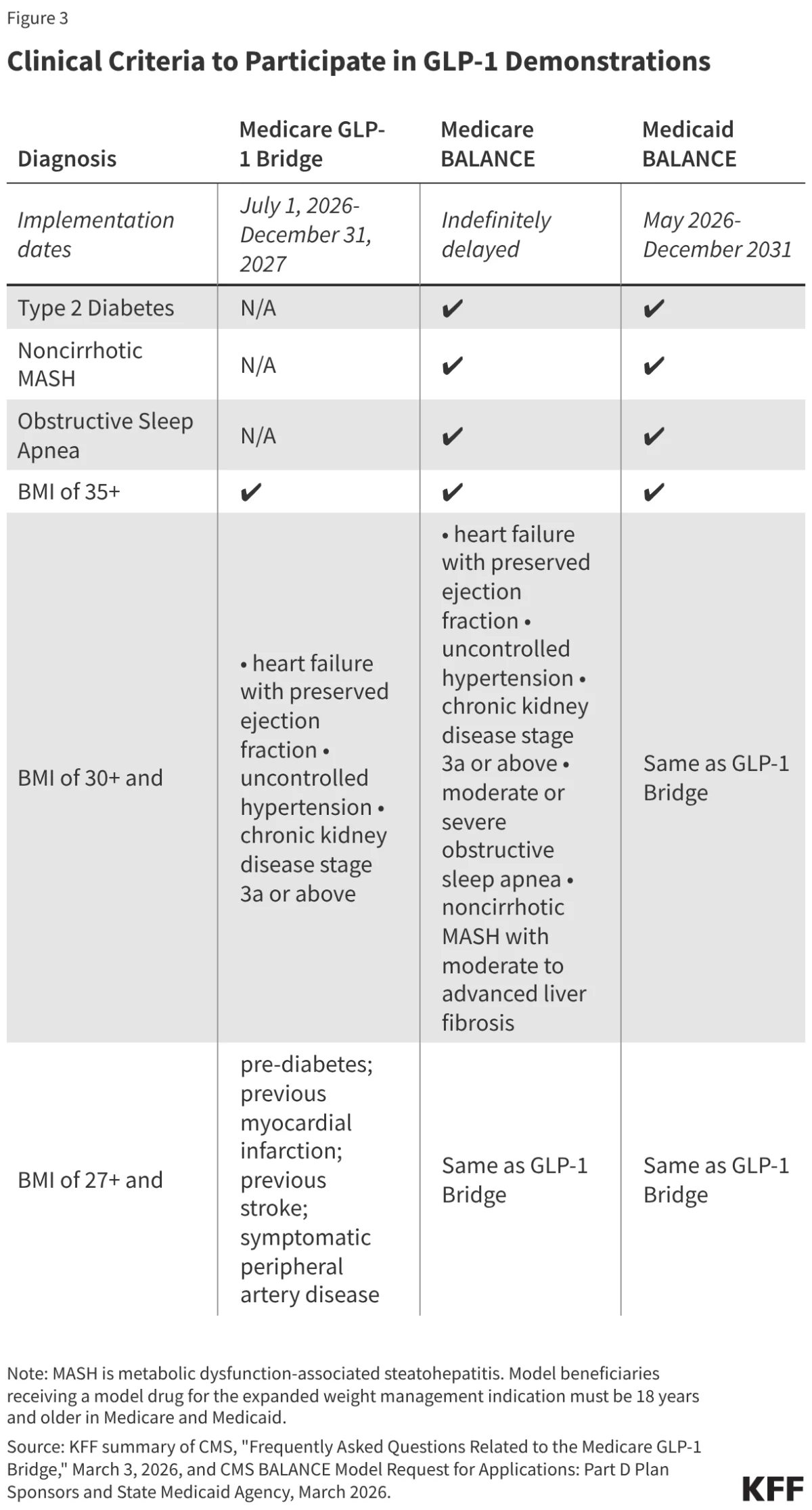
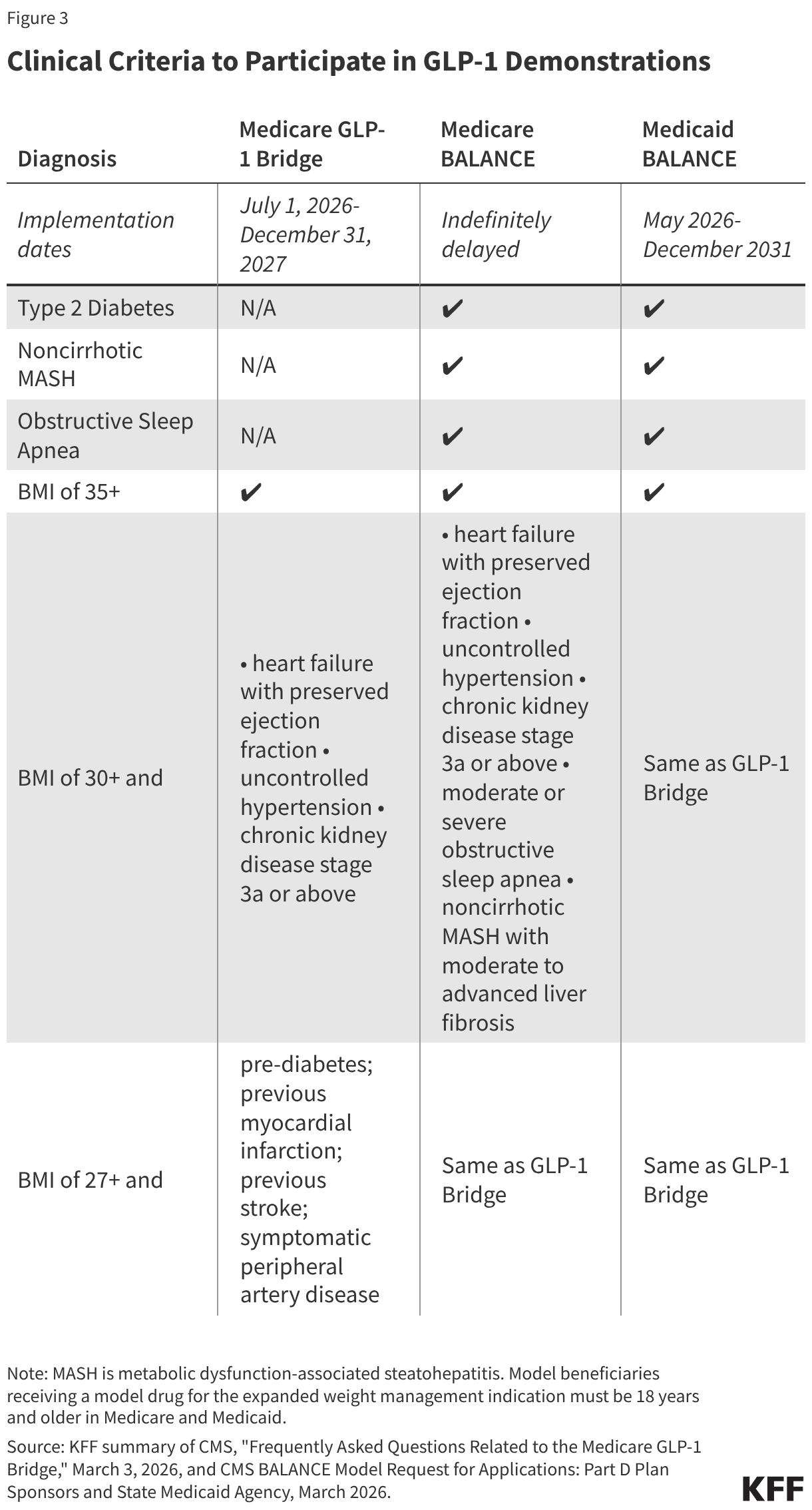
To bridge the gap until a more permanent policy is realized, CMS introduced the Medicare GLP-1 Bridge. This nationwide demonstration, utilizing Section 402 authority, provides a pathway for Part D beneficiaries to access select GLP-1s specifically for weight reduction.
Key Operational Features:
- Cost and Access: Beneficiaries pay a flat $50 monthly copayment. Pharmacies are reimbursed by CMS at the wholesale acquisition cost (WAC), while manufacturers provide a rebate to bring the net cost down to $245 per 30-day supply.
- Exclusion from Part D: Crucially, this program operates outside the standard Part D benefit. Consequently, the $50 copayments do not count toward the beneficiary’s annual out-of-pocket maximum ($2,100 in 2026; $2,400 in 2027).
- Eligibility: To qualify, providers must submit a prior authorization attesting that the medication is for weight reduction and maintenance, meeting specific BMI and clinical diagnostic thresholds.
While the Bridge program provides essential relief, its temporary nature and the exclusion of the Low-Income Subsidy (LIS) cost-sharing protections raise concerns about affordability for the most vulnerable seniors.
The BALANCE Model: Design and Disappointment
The BALANCE Model was conceived as a comprehensive solution, integrating pharmaceutical cost reduction with mandatory lifestyle support programs. By negotiating lower net prices with manufacturers and requiring that they provide healthy eating and physical activity resources, CMS hoped to move toward a value-based care model.
However, the "critical mass" required for the model’s success—an 80% participation rate among Part D plans—failed to materialize. Reports suggest that major plan sponsors were hesitant to bear the financial risk associated with a high-volume, high-cost drug class in a program that lacked robust long-term budgetary assurances. While the model proceeds in the Medicaid space, its failure to launch in Medicare highlights the tension between public health goals and the private market’s risk-averse nature.
Implications: The Road Ahead
The indefinite postponement of the BALANCE Model in Medicare creates a state of policy uncertainty. For the millions of Americans struggling with obesity, the current strategy is a patch, not a foundation.
Financial Impacts
The budgetary implications for the federal government remain opaque. While CMS aims to lower prices through negotiation, the potential surge in utilization—combined with the high list price of GLP-1s—could place immense pressure on the federal budget. Estimates suggest that covering obesity drugs in Medicare could cost between $25 billion and $35 billion over a decade. Whether the cost savings from improved health outcomes can eventually offset these expenditures remains a subject of intense debate among health economists.
Potential for Coverage Gaps
There is a looming risk of "coverage cliffs." If the GLP-1 Bridge program ends in 2027 and no follow-up program is established, beneficiaries could face a sudden loss of access to their medications. Furthermore, if the BALANCE Model eventually restarts, frequent changes in plan participation could lead to disruptions in patient care, forcing enrollees to navigate shifting formularies and provider networks to maintain their treatments.
The Role of Data and Evidence
CMS has indicated that the delay in the BALANCE Model for Medicare is partly designed to allow for the collection of more comprehensive utilization data. This information will be vital for plan sponsors to accurately price their bids in future years. However, the lack of real-world evidence confirming that short-term GLP-1 use leads to immediate, cost-offsetting health improvements means that the long-term fiscal viability of these programs remains speculative.
Ultimately, the administration’s current approach underscores a fundamental conflict: the desire to provide high-cost, high-value medical innovations to a vast population without dismantling the financial solvency of the existing Medicare Part D structure. Until a permanent legislative or regulatory consensus is reached, beneficiaries are left in a precarious position, dependent on temporary demonstrations that may change with the political and economic tides.