By Science Correspondent
Peripheral Arterial Disease (PAD) has long been described as a "silent" epidemic. Affecting an estimated 200 million people worldwide, this chronic atherosclerotic condition—characterized by the narrowing of arteries in the lower extremities—often remains undiagnosed until it reaches a dangerous, advanced stage. However, a groundbreaking study published in the journal Diagnostics suggests that a simple blood-based biomarker, Fatty Acid Binding Protein 3 (FABP3), may finally provide the diagnostic precision clinicians have been seeking.
Led by Dr. Mohammad Qadura of Unity Health Toronto, a team of researchers has demonstrated that integrating FABP3 levels with standard clinical risk factors significantly improves the detection of PAD, particularly in asymptomatic patients who previously slipped through the cracks of traditional screening methods.

The Persistent Challenge of PAD Diagnosis
PAD is more than just leg pain; it is a systemic marker of cardiovascular health, linked to severe morbidity, limb loss, and increased all-cause mortality. Currently, the medical community relies heavily on the Ankle-Brachial Index (ABI)—a non-invasive test that compares blood pressure at the ankle to that in the arm.
While the ABI is a staple of vascular surgery clinics, it is fundamentally flawed. In patients with diabetes or chronic kidney disease, calcification of the arterial walls can cause "non-compressible" vessels, leading to falsely elevated readings that mask the true severity of the disease. Furthermore, the ABI is notoriously ineffective at identifying early-stage or asymptomatic PAD. Consequently, many patients only receive a diagnosis after they have developed critical limb-threatening ischemia, leaving them with limited, often invasive treatment options.
"There has been a conspicuous void in specific biomarkers tailored for PAD," explains the research team. While fields like cardiology have benefited from sensitive blood markers like troponins for myocardial infarction or NT-proBNP for heart failure, vascular medicine has lacked a reliable "molecular flag" for arterial damage.

A New "Ischemia Sensor": The Role of FABP3
Fatty Acid Binding Protein 3 (FABP3) is a low-molecular-weight protein expressed in cardiac and skeletal muscle. Under normal physiological conditions, it manages the transport of fatty acids to meet the high energy demands of muscle tissue. However, when muscle fibers are subjected to the hypoxia and stress caused by arterial narrowing, FABP3 leaks into the bloodstream.
Researchers hypothesize that FABP3 acts as an "ischemia sensor," signaling damage at the cellular level long before anatomical changes become visible on traditional imaging like duplex ultrasound or CT angiography. By measuring these circulating levels, clinicians could potentially identify PAD at a stage where lifestyle interventions and early medical management can prevent future complications.
Chronology of the Study: A Rigorous Retrospective Analysis
The study, which was published on May 11, 2026, represents the culmination of years of clinical data collection.

- February 4, 2026: The research was officially submitted for peer review, detailing a retrospective analysis of 657 patients who presented to ambulatory vascular surgery clinics at St. Michael’s Hospital in Toronto.
- October 2017 – July 2019: The original patient cohort was recruited, ensuring a comprehensive data set that included ABI testing, physical examinations, and banked plasma samples.
- April 6, 2026: Following a thorough peer-review process, the manuscript was revised to address methodological nuances, including the strict exclusion of patients with recent acute coronary syndromes or renal failure to ensure that elevated FABP3 levels could be confidently linked to peripheral ischemia.
- April 20, 2026: The study was accepted for publication, confirming the statistical significance of the findings.
- May 11, 2026: Official publication in Diagnostics.
Supporting Data: Outperforming Traditional Methods
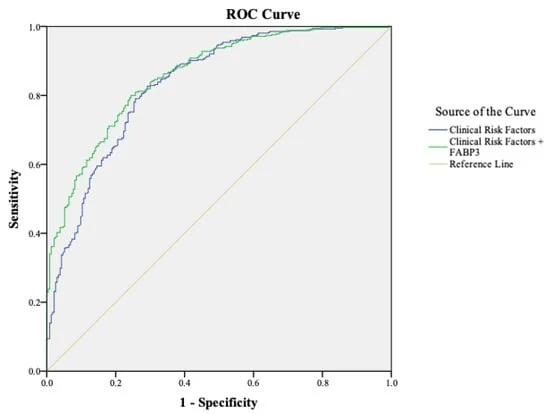
The research team compared two distinct logistic regression models. Model A relied on traditional clinical risk factors—age, smoking, diabetes, hypertension, and known coronary artery disease. Model B integrated these same factors with plasma FABP3 levels.
The results were striking:
- Diagnostic Accuracy: Model A achieved a diagnostic accuracy of 76%. With the integration of FABP3, Model B reached 93%.
- Sensitivity and Specificity: The FABP3-integrated model boasted a 96% sensitivity and 84% specificity, compared to 81% and 67%, respectively, for the traditional model.
- The "Silent" Population: Perhaps most significantly, Model A failed to detect all 82 asymptomatic PAD cases in the cohort. In contrast, Model B successfully identified 84% of these "silent" cases.
The study also utilized the Net Reclassification Improvement (NRI) metric, which confirmed a significant 29% improvement in patient classification accuracy, proving that FABP3 provides actionable clinical information beyond what can be determined by a physical examination alone.

Implications for Clinical Practice
The integration of a blood test into routine screening could fundamentally shift the paradigm of vascular care.
1. Reaching the "Grey Zone"
A significant portion of patients—277 individuals in the study—fell into a "grey zone" where diagnostic ambiguity was high. In these cases, where the ABI was inconclusive, the addition of FABP3 helped clarify the clinical picture, offering physicians a clear path forward for treatment.
2. Early Detection and Prevention
By identifying PAD before it progresses to claudication or gangrene, clinicians can implement aggressive lipid-lowering therapies, antiplatelet medications, and smoking cessation programs. This preventative approach is expected to reduce the long-term burden on healthcare systems and, more importantly, preserve the quality of life for millions of patients.

3. A Complement, Not a Replacement
The researchers emphasize that FABP3 is not intended to replace existing diagnostic tools like the ABI or ultrasound. Instead, it is a complementary biomarker. In high-risk groups, such as those with diabetes, FABP3 could act as a "gatekeeper" test, flagging those who require immediate advanced imaging while sparing others from unnecessary, expensive, and time-consuming diagnostic procedures.
Official Perspective and Future Outlook
While the results are highly promising, the authors urge caution. The study was a single-center retrospective analysis, meaning that larger, multi-center, prospective trials are necessary to validate these findings across diverse populations.
"FABP3 represents a promising and practical biomarker for PAD," the authors concluded in their final report. "While additional prospective validation is essential, our findings mark a critical step toward biomarker-driven precision vascular care."

The study was supported by funding from The Bill and Vicky Blair Foundation and strictly adhered to the Declaration of Helsinki, with all participant data handled according to established ethical guidelines at Unity Health Toronto.
As the medical community continues to look for ways to manage the rising prevalence of chronic vascular diseases, the "silent" nature of PAD may finally be ending. By looking at the blood for molecular markers of ischemia, doctors may soon be able to detect this dangerous condition with the same ease and precision as they do a heart attack, potentially saving countless limbs—and lives—in the process.