Introduction
Gonadal germ cell tumors (GCTs), originating from primordial germ cells within the testes and ovaries, represent a fascinating spectrum of neoplastic entities. While sharing a common embryological origin, these tumors exhibit striking differences in incidence, histological subtypes, and, crucially, their underlying molecular and genetic landscapes. Understanding these nuances is paramount for accurate classification, the development of novel biomarkers, and the refinement of therapeutic strategies. This comprehensive review delves into the comparative molecular and genetic landscape of testicular and ovarian GCTs, synthesizing current evidence to illuminate their shared origins and divergent evolutionary paths.
The Genesis of Germ Cell Tumors: A Shared Blueprint with Divergent Execution
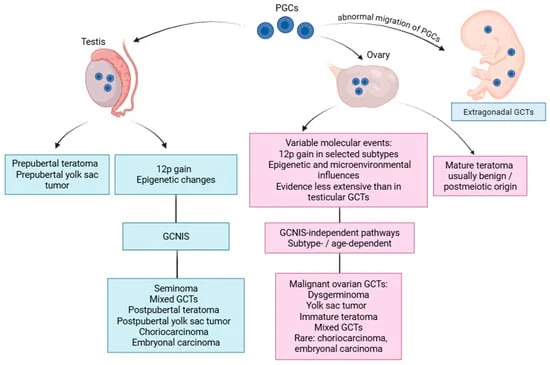
At the heart of all GCTs lies the primordial germ cell (PGC), a pluripotent cell population that embarks on a complex migratory journey from the yolk sac to the developing genital ridges. This fundamental migratory pathway, governed by intricate molecular cues, provides a unifying explanation for the occurrence of GCTs in both gonadal sites and, occasionally, in extragonadal locations along the embryonic midline.
The journey of PGCs is not without peril. Disruptions in their specification, migration, or colonization of the genital ridge can predispose to tumorigenesis. This concept positions GCTs as developmental disorders rather than conventional malignancies, a perspective further supported by the identification of dysregulated pathways crucial for early germ cell development. The PI3K/PTEN/AKT pathway, a critical regulator of cell survival, proliferation, and metabolism, has emerged as a significant player. Its dysregulation in GCTs can fuel abnormal cell growth and contribute to the maintenance of a stem-like phenotype, a characteristic observed in these tumors.
However, the gonadal microenvironment plays a pivotal role in dictating the fate of these germ cells. Upon reaching the developing testis or ovary, PGCs are influenced by somatic support cells, hormonal signals, and the overall developmental context. This interaction leads to divergent pathways: spermatogenesis in males and oogenesis in females. It is within this gonadal-specific milieu that GCTs also diverge. While testicular GCTs have a well-defined precursor lesion, germ cell neoplasia in situ (GCNIS), a definitive ovarian counterpart remains elusive, suggesting distinct routes to malignant transformation.
Epidemiology and Histological Nuances: A Tale of Two Gonads

The epidemiological profiles of testicular and ovarian GCTs offer the first glimpse into their divergent nature. Testicular GCTs are the most common solid tumors in young adult males, with a peak incidence in the second and third decades of life. Conversely, ovarian GCTs, while less common overall, are frequently encountered in adolescents and young adults, with some subtypes showing an earlier age of onset compared to their testicular counterparts.
Histologically, there is overlap, with entities like teratoma, seminoma/dysgerminoma, yolk sac tumor, and mixed germ cell tumors recognized in both sexes. However, subtle yet significant differences exist. Testicular teratomas are classified as prepubertal (Type I) or postpubertal (Type II), with the latter arising from GCNIS. Ovarian teratomas, predominantly mature forms, are the most frequent GCTs, while immature teratomas also occur. Testicular GCTs are also more commonly associated with the presence of GCNIS, a pre-invasive lesion that serves as a crucial precursor for many malignant testicular tumors. The absence of a well-established equivalent in the ovary underscores a fundamental biological distinction. Furthermore, certain rare histological subtypes, such as spermatocytic tumor, are exclusive to the testis, highlighting site-specific tumorigenesis.
Comparative Molecular and Genetic Landscape: Unpacking the Divergence
Cytogenetic Profile: The Chromosome 12p Signature and Beyond
Cytogenetic analysis has revealed both shared and distinct chromosomal abnormalities in gonadal GCTs. A hallmark of postpubertal testicular GCTs, particularly Type II tumors, is the gain of chromosome 12p, most commonly manifesting as an isochromosome 12p (i(12p)). This alteration is present in over 80% of these tumors and is strongly associated with tumor invasiveness and progression from GCNIS.
Ovarian GCTs also exhibit chromosomal aberrations, but their profile is more heterogeneous and subtype-dependent. While 12p gains have been identified in subsets of malignant ovarian GCTs, they are not as consistently prevalent or as mechanistically linked to a specific precursor lesion as in the testis. Instead, ovarian GCTs show a broader range of copy-number alterations, including gains and losses across various chromosomes, with some regions, like 1p36, being recurrently deleted. This suggests that while chromosome 12p may play a role in germ cell tumorigenesis across gonadal sites, its significance and the overall cytogenetic landscape differ considerably.
Epigenetic Landscape: Shared Germ Cell Signatures and Gonad-Specific Programs
Epigenetic dysregulation plays a pivotal role in the biology of GCTs, often compensating for a relatively low somatic mutation burden. Both testicular and ovarian GCTs share conserved epigenetic features reflective of their primordial germ cell origin. These include global DNA hypomethylation, characteristic microRNA expression profiles, and the preservation of pluripotency-associated epigenetic programs.
The miR-371~373 and miR-302/367 clusters, for instance, are consistently overexpressed across malignant GCTs, regardless of gonadal origin. MicroRNA miR-371a-3p, in particular, has emerged as a promising circulating biomarker for diagnosis and monitoring of malignant GCTs, demonstrating high sensitivity and specificity. However, its clinical validation is strongest in testicular GCTs, with more limited data available for ovarian counterparts.

Despite these shared signatures, gonad-specific epigenetic trajectories are evident. Ovarian teratomas, for example, display distinct imprinting and methylation patterns consistent with a parthenogenetic origin, a phenomenon not observed in testicular tumors. Furthermore, the association between DNA methylation patterns and cisplatin sensitivity, a well-established feature of testicular GCTs, appears less clearly defined in ovarian tumors, highlighting the influence of the gonadal microenvironment on epigenetic regulation and therapeutic response.
Mutational Profiles: Low Burden, Targeted Pathways, and Emerging Differences
Compared to many common adult solid tumors, testicular and ovarian GCTs generally exhibit a low burden of recurrent somatic point mutations. However, the mutations that do occur are often biologically significant and can illuminate key oncogenic pathways.
In testicular GCTs, activating mutations in KIT and RAS pathway genes (KRAS, NRAS) are among the most frequently observed somatic point mutations, particularly in seminoma subtypes. These alterations drive proliferative signaling via MAPK pathways and are implicated in tumor progression. Somatic mutations in TP53, while rare in treatment-naive tumors, are emerging as a hallmark of resistance to platinum-based chemotherapy, suggesting a role in adaptive chemoresistance.
Ovarian GCTs, in contrast, display a more heterogeneous and less consistent mutational landscape. While a low overall mutation rate is observed, some studies suggest potential alterations in the PI3K/AKT/PTEN pathway in yolk sac tumor components and rare mutations in genes like POU5F1, which is involved in pluripotency networks. However, the limited size of many ovarian GCT cohorts and the dominance of chromosomal aberrations over point mutations make it challenging to draw definitive conclusions about recurrent drivers.
The comparative analysis reveals that while both GCT types share a low mutation rate, they diverge in the specific somatic events that recur and shape their biology. Testicular GCTs exhibit more defined mutational hotspots, whereas ovarian GCTs reflect greater heterogeneity and a lower recurrence of specific point mutations.
Integrative Insights and Future Directions: Bridging the Gap
The comparative analysis of testicular and ovarian GCTs underscores the profound influence of the gonadal microenvironment on tumor development and behavior. While a common germ cell origin provides a unified molecular framework, the distinct somatic support, hormonal milieu, and developmental timing within the testis and ovary lead to divergent evolutionary pathways.

The remarkable sensitivity of most GCTs to platinum-based chemotherapy is a testament to their shared developmental and epigenetic underpinnings. However, the emergence of resistance in a subset of patients highlights the need for a deeper understanding of the molecular determinants of treatment response. Identifying these determinants could pave the way for more precise risk stratification and the development of novel therapeutic strategies for refractory disease.
Emerging research on the tumor microenvironment suggests that immune cells and checkpoint molecules may also play a role in GCT biology. While immune checkpoint inhibitors have shown limited efficacy in heavily pretreated GCTs, further investigation into the complex interplay between tumors and the immune system is warranted.
Limitations and Strength of Evidence
It is crucial to acknowledge the inherent asymmetry in the literature concerning gonadal GCTs. Testicular GCTs have been more extensively studied at the molecular and genetic levels, providing a robust dataset for comparison. Ovarian GCTs, being rarer and more histologically diverse, have smaller cohorts and less comprehensive profiling data. Consequently, some comparative conclusions are well-supported by extensive evidence, while others remain provisional and require further validation. Careful distinction between established shared mechanisms and emerging observations is therefore essential for an accurate interpretation of the current scientific landscape.
Conclusion: Unity and Divergence in Germ Cell Neoplasms
Gonadal germ cell tumors, despite their shared origin from primordial germ cells, present a compelling case study in how developmental context shapes neoplastic evolution. While sharing fundamental cytogenetic hallmarks like chromosome 12p gain in postpubertal tumors and conserved epigenetic signatures, testicular and ovarian GCTs diverge significantly in their precursor lesions, the frequency and biological relevance of specific molecular alterations, and their overall clinical behavior.
A nuanced understanding of both the unifying pathways and the site-specific divergences is critical for advancing the field. Future research, leveraging high-resolution multi-omics profiling and single-cell technologies across both gonadal contexts, holds the promise of unraveling the intricate mechanisms driving germ cell tumorigenesis. Such integrative approaches will not only refine our classification and diagnostic capabilities but also pave the way for the development of more personalized and effective therapeutic strategies for patients diagnosed with these complex neoplasms.