By [Your Name/Journalistic Desk]
May 10, 2026
The future of medication abortion in the United States hangs in a precarious legal balance as the Supreme Court prepares to intervene in Louisiana v. FDA. Following a volatile week of emergency filings, the nation’s highest court is now tasked with determining whether a restrictive Fifth Circuit ruling will stand, potentially upending the current nationwide access to mifepristone.
Main Facts: The Current Constitutional and Regulatory Conflict
At the center of the controversy is the U.S. Food and Drug Administration’s (FDA) 2023 policy, which modernized access to mifepristone by removing the long-standing requirement that the drug be dispensed in person. This change allowed for telehealth consultations and the distribution of the medication via mail and retail pharmacies.
On May 4, 2026, Justice Samuel Alito issued a one-week administrative stay of a May 1st ruling by the U.S. Court of Appeals for the Fifth Circuit. That appellate decision had temporarily reinstated the in-person dispensing requirement, a move that would have effectively barred the mailing of abortion pills nationwide. The Supreme Court’s intervention ensures that, for at least one week, the current FDA protocols remain in place while the Justices evaluate the emergency appeals filed by the drug’s manufacturers, Danco Laboratories and GenBioPro.
This case is not merely about a single drug; it is a fundamental clash between federal administrative authority and state police power. The central question is whether a state can claim "legal standing"—the right to sue—by arguing that federal drug policies cause the state economic harm, specifically through Medicaid expenditures on patients who require emergency care following a medication abortion.
Chronology of a Mounting Legal Crisis
- 2000: The FDA initially approves mifepristone with a strict Risk Evaluation and Mitigation Strategy (REMS), mandating in-person physician dispensing.
- 2023: After a comprehensive review of safety data, the FDA formally eliminates the in-person requirement, facilitating mail-order access and pharmacy distribution.
- 2024: The Supreme Court rules in Alliance for Hippocratic Medicine v. FDA that anti-abortion plaintiffs lacked standing, avoiding a decision on the drug’s safety or the legality of the FDA’s regulatory process.
- October 2025: The State of Louisiana sues the FDA, alleging that the 2023 REMS violates the Administrative Procedure Act (APA) and the 1873 Comstock Act.
- April 2026: A federal district court pauses the litigation for six months to allow the FDA to complete an internal safety review. Louisiana appeals this pause to the Fifth Circuit.
- May 1, 2026: The Fifth Circuit panel grants Louisiana’s request, ordering an immediate return to in-person dispensing.
- May 4, 2026: Justice Alito grants an emergency administrative stay, halting the Fifth Circuit’s order until May 11, 2026.
Supporting Data: Safety and Access Metrics
The debate over mifepristone is frequently colored by diverging interpretations of safety data. Medication abortion, typically a two-drug regimen of mifepristone and misoprostol, has become the most common method of abortion in the U.S.
Data from the #WeCount project of the Society of Family Planning indicates that as of June 2025, approximately 55% of all telehealth medication abortions were provided to patients residing in states with total or significant abortion bans. This underscores the reliance on mail-order services to bypass state-level restrictions.
Regarding safety, the FDA’s 2023 decision was based on over two decades of clinical evidence, including:
- A 2021 literature review of published studies.
- Five years of adverse event data.
- A specialized one-year assessment report specifically for the REMS.
The FDA concluded that removing the in-person requirement did not introduce new safety concerns and that the benefits of the drug continued to outweigh the risks. However, opponents, including Louisiana officials, cite specific cases—such as $92,000 in Medicaid costs incurred by two patients in 2025—to argue that the federal government is shifting the financial burden of complications onto state taxpayers.
Official Responses: The "Tightrope" of the FDA
The position of the FDA has shifted significantly under the current administration. While the agency continues to defend its regulatory authority in court, the executive branch has signaled a change in philosophy. In September 2025, the FDA announced a new, comprehensive review of the 2023 REMS, citing "recent studies raising concerns about the safety of mifepristone."
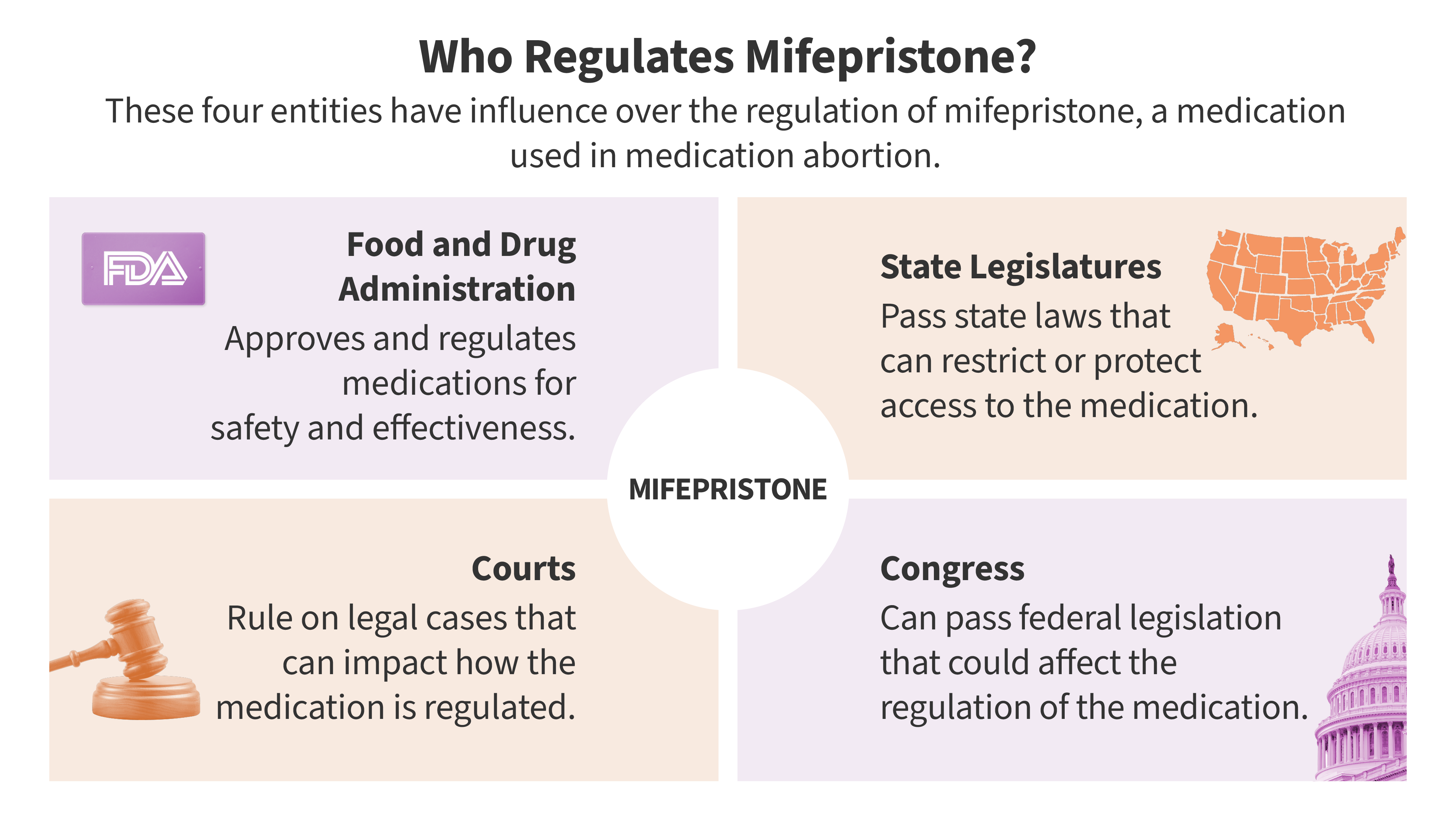
This acknowledgment has become a double-edged sword. The Fifth Circuit used the FDA’s own admission of a "review" to suggest the agency conceded that its previous removal of the in-person requirement might have been procedurally flawed under the APA. Conversely, the drug manufacturers argue that the FDA is being pressured by political actors to undermine its own established scientific record.
Senator Josh Hawley (R-MO) and others have intensified this pressure by launching congressional investigations into Danco and GenBioPro, demanding data on adverse events and questioning the manufacturers’ trade practices.
Implications: Federal Preemption and Interstate Conflict
The ripple effects of this litigation extend far beyond the pharmacy counter.
1. The Challenge to Federal Supremacy
If the Supreme Court rules that Louisiana has standing, it could set a dangerous precedent. The defendants—the FDA and the manufacturers—argue that if a state can sue the federal government over every drug that leads to a hospital visit, the administrative state would be paralyzed by endless litigation. They cite the Supreme Court’s own logic in the Alliance case, where the Court warned against allowing states to challenge federal policies simply because of indirect, downstream economic consequences.
2. The Rise of "Shield Laws"
A significant development in the post-Dobbs landscape is the enactment of "shield laws" in states like California and New York. These laws protect telehealth providers from extradition and prosecution by states like Texas or Louisiana that seek to criminalize the mailing of abortion pills across state lines. This has created a fractured national landscape, where the legality of a medical procedure depends entirely on the geographical location of the provider versus the patient.
3. The "Misoprostol-Only" Alternative
Should the Supreme Court allow the Fifth Circuit’s order to take effect, the medical community is bracing for a shift. Many clinics are prepared to pivot to a misoprostol-only protocol. While this regimen is a recognized alternative used globally, it is generally considered less effective (roughly 80-100% depending on the protocol) than the combination of mifepristone and misoprostol (91.9-99.7%) and is associated with higher rates of pain and gastrointestinal side effects.
4. The Potential for Unprecedented Judicial Intervention
Legal scholars note that it is virtually unprecedented for a federal court to compel the FDA to modify a REMS for an approved medication. If the courts begin to dictate specific safety protocols for drugs—bypassing the scientific expertise of the FDA—it could destabilize the entire framework of the U.S. pharmaceutical industry.
Looking Ahead
As the May 11th expiration of the stay approaches, the Supreme Court faces a defining moment. A decision to uphold the Fifth Circuit’s injunction would be a major victory for states seeking to restrict abortion, effectively ending the era of remote medication abortion. A decision to reject the Fifth Circuit’s reasoning would reaffirm the FDA’s role as the primary regulator of drug safety.
Ultimately, Louisiana v. FDA is the latest, and perhaps most complex, chapter in a post-Roe era where the line between state sovereignty and federal authority is being redrawn in the courtrooms. The outcome will not only impact millions of patients seeking reproductive healthcare but will also redefine the boundaries of federal regulatory power for decades to come.