[City, Date] – In a significant development for the treatment of transthyretin amyloid cardiomyopathy (ATTR-CM), a rare and progressive cardiac disease, BridgeBio Pharma has unveiled compelling new data for its therapy, Attruby (acoramidis). The findings, presented at the European Society of Cardiology’s (ESC) 2026 Heart Failure conference, not only reinforce Attruby’s efficacy and safety but also suggest its potential to outperform Pfizer’s established treatment, Vyndamax (tafamidis), in frontline therapy, based on an indirect comparison.
This latest data, derived from the pivotal Phase III ATTRibute-CM study, positions Attruby as a strong emerging force in a market currently dominated by tafamidis. The US Food and Drug Administration (FDA) granted approval for Attruby in November 2024, marking a critical milestone for patients and for BridgeBio’s ambition to capture substantial market share in ATTR-CM.
Attruby’s Mechanism of Action and Clinical Efficacy
ATTR-CM is caused by the misfolding and aggregation of the transthyretin (TTR) protein, which forms amyloid fibrils that deposit in the heart muscle, leading to progressive thickening and stiffening. Attruby’s therapeutic strategy focuses on stabilizing the TTR protein in its native, tetrameric form, thereby preventing its dissociation and subsequent misfolding.
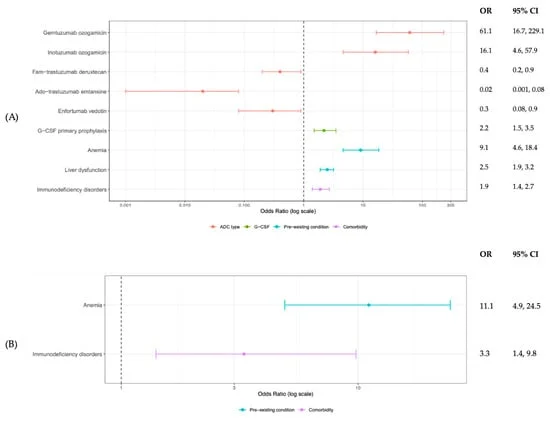
The Phase III ATTRibute-CM study (NCT03860935) evaluated the efficacy and safety of Attruby in patients with ATTR-CM. Key findings presented highlighted the drug’s ability to significantly boost and maintain levels of wild-type transthyretin from day 28 through to month 30 of treatment. This sustained stabilization of the TTR protein is crucial for halting the progression of the disease.
Furthermore, patients treated with Attruby experienced a remarkable reduction in the variability of their blood transthyretin levels compared to placebo. This reduction in variability was linked to a significant positive impact on the risk of all-cause mortality, mediating approximately 20% of Attruby’s observed benefit.
Dr. Senthil Selvaraj, from Duke University School of Medicine, emphasized the mechanistic implications of these findings. "By linking transthyretin variability independently to mortality, we’re seeing a mechanistic signal that may help explain Attruby’s clinical benefit," Dr. Selvaraj stated. "Reducing transthyretin variability on an individual level may be important in modifying disease outcomes."
In addition to its impact on mortality, data presented by Bayer, BridgeBio’s European licensing partner, who markets the drug as Beyonttra, demonstrated Attruby’s potential to improve patient outcomes by diminishing the risk of outpatient worsening heart failure by 40% versus placebo. This is a critical indicator, as worsening heart failure is a strong predictor of mortality and hospitalization in ATTR-CM patients.
Indirect Comparison: Attruby’s Potential to Surpass Current Standard of Care
Perhaps the most striking revelation from the ESC conference was the outcome of a matching-adjusted indirect comparison (MAIC) that pitted Attruby against Pfizer’s Vyndamax (tafamidis), the current gold standard and mainstay therapy for ATTR-CM. In Europe, Vyndamax is marketed as Vyndaqel.
The MAIC, drawing data from the ATTRibute-CM study and relevant trials of tafamidis, indicated that Attruby could offer superior patient outcomes in frontline ATTR-CM. The comparison revealed a significant 34% reduction in cardiovascular hospitalizations for Attruby compared to tafamidis, while maintaining a comparable safety profile. This suggests that Attruby may be more effective in preventing the acute cardiac events that frequently complicate ATTR-CM.
Moreover, Attruby demonstrated a favorable mortality trend, exhibiting a 28% hazard reduction in all-cause mortality compared to tafamidis. While these results are highly encouraging, it is crucial to note that BridgeBio has not conducted a direct head-to-head study against Vyndamax. Therefore, definitive conclusions on the direct comparability between the two drugs cannot yet be drawn. Nevertheless, the indirect comparison provides a strong signal of Attruby’s potential to offer an improved therapeutic profile.
Chronology of Attruby’s Development and Market Entry
The journey of Attruby from research to clinical application has been marked by significant milestones:
- Pre-clinical Research and Early Development: BridgeBio has been actively developing acoramidis, focusing on its unique TTR-stabilizing mechanism.
- Phase III ATTRibute-CM Study Initiation: The pivotal ATTRibute-CM trial (NCT03860935) was initiated to rigorously evaluate the drug’s efficacy and safety in patients with ATTR-CM.
- European Licensing Agreement: BridgeBio partnered with Bayer for the European licensing and commercialization of acoramidis, which is marketed as Beyonttra in this region.
- FDA Approval: In November 2024, the US Food and Drug Administration (FDA) granted approval for Attruby for the treatment of ATTR-CM, marking a major regulatory success.
- ESC 2026 Heart Failure Conference Presentation: New pivotal trial data from ATTRibute-CM and the indirect comparison to tafamidis were presented, generating significant attention within the cardiology community.
Supporting Data and Market Performance
The promising clinical data for Attruby is already translating into strong market performance. The drug has surpassed analyst expectations, generating $180.6 million in first-quarter revenues, a substantial 24% increase from the same quarter in 2025. This robust revenue growth underscores Attruby’s rapid adoption and increasing market share.
Analysts at William Blair noted the alignment of fundamental factors driving Attruby’s success. Their research indicates that Attruby is capturing a significant and growing percentage of first-line usage, alongside meaningful second-line usage despite sharing a similar mechanism of action with first-line Vyndamax. Furthermore, its combination usage with knockdown therapies is approaching parity with Vyndamax, suggesting a broadening therapeutic role.
Official Responses and Industry Reactions
The presentation of Attruby’s data at the ESC conference has been met with considerable interest from the medical and pharmaceutical communities. While specific official statements from Pfizer regarding the indirect comparison were not immediately available, the market dynamics suggest an evolving competitive landscape.
The indirect comparison data, in particular, will likely prompt further investigation and potentially influence prescribing patterns. The fact that Attruby is showing potential to improve upon the outcomes achieved by tafamidis, without a head-to-head trial, speaks to the strength of the ATTRibute-CM data and the robustness of the MAIC methodology employed.
Implications for the ATTR-CM Market and Future Outlook
The emergence of Attruby as a potential superior frontline therapy for ATTR-CM carries significant implications:
- Increased Competition and Patient Choice: The growing efficacy of Attruby will likely intensify competition in the ATTR-CM market, offering physicians and patients more therapeutic options. This can lead to better treatment tailored to individual patient needs.
- Shifting Market Dynamics: If Attruby continues to demonstrate its advantages in real-world settings, it could lead to a substantial shift in market share away from existing therapies, including Vyndamax.
- Focus on Novel Mechanisms: The success of Attruby, which utilizes a TTR stabilization approach, reinforces the value of targeting the underlying disease mechanism. This may encourage further investment in similar therapeutic strategies.
- Impact of Patent Expiries: Pfizer’s recent settlements with several pharmaceutical companies regarding Vyndamax generics, pushing its US patent expiry to 2031, represent a mixed outcome for BridgeBio. While this delays the entry of generic competition for Pfizer, it also extends the period during which Attruby must compete against a branded, established therapy. However, the potential for Attruby to offer superior outcomes could still allow it to carve out a significant market share even with the extended patent life of Vyndamax.
- Pipeline Development: Currently, Attruby, Vyndamax, and Alnylam Pharmaceuticals’ Amvuttra (vutrisiran) are the only US-approved drugs for ATTR-CM. However, the pipeline remains active, with therapies from Ionis Pharmaceuticals, Novo Nordisk, and Intellia Therapeutics progressing through late-stage development. Attruby’s strong performance could influence the strategic positioning of these emerging therapies.
The future of ATTR-CM treatment appears increasingly dynamic, with Attruby poised to play a pivotal role. The robust data presented at the ESC conference, particularly the indirect comparison suggesting advantages over tafamidis, signals a potential paradigm shift in frontline therapy. As BridgeBio continues to establish its market presence and further real-world data emerges, Attruby is well-positioned to become a cornerstone in the management of this devastating cardiac condition.