A comprehensive analysis of over 3,500 patients treated across six University of California medical centers has provided crucial insights into the incidence and severity of neutropenia-related complications associated with antibody-drug conjugates (ADCs), a rapidly evolving class of targeted cancer therapies. The study, published in the journal Cancers, highlights significant variations in the risk of low white blood cell counts and associated infections among different ADCs, underscoring the need for tailored monitoring and supportive care strategies.
Main Facts: Uneven Safety Landscape for ADCs
The extensive real-world data analysis, encompassing 3,511 patients treated between 2012 and 2024, revealed that while some ADCs are associated with a relatively low risk of serious neutropenic events, others carry a substantially higher burden. Key findings include:

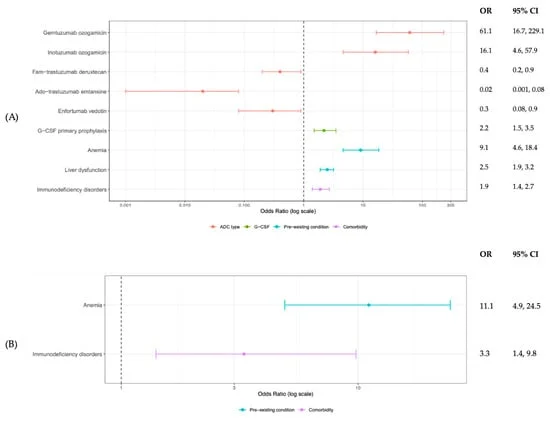
- Variable Risk Profiles: ADCs like fam-trastuzumab deruxtecan and ado-trastuzumab emtansine demonstrated lower rates of febrile neutropenia (FN) and grade 3 or higher neutropenia compared to other agents.
- Higher Risk in Hematologic Malignancies: ADCs primarily used to treat blood cancers, such as gemtuzumab ozogamicin and inotuzumab ozogamicin, were linked to significantly higher incidences of severe neutropenia and subsequent complications like hospitalizations and intensive care unit (ICU) admissions.
- Predictive Factors Identified: Pre-existing anemia emerged as a strong predictor for neutropenic events across various ADC classes and cancer types. Other contributing factors included liver dysfunction, immunodeficiency, and the use of growth factor prophylaxis.
- Healthcare Utilization: The study detailed varying rates of G-CSF prophylaxis, hospital admissions, ICU admissions, and mortality, correlating with the identified neutropenic risks of specific ADCs.
Chronology: A Decade of Real-World ADC Use Under Scrutiny
The research meticulously examined a decade of patient data, from January 2012 to July 2024, providing a historical perspective on the real-world application of the ten most frequently prescribed ADCs. This longitudinal approach allowed researchers to capture a broad spectrum of treatment patterns and outcomes as the use of these innovative therapies expanded. The retrospective nature of the study, utilizing the robust University of California Health Data Warehouse, provided a unique opportunity to analyze outcomes in a diverse patient population representative of routine clinical practice, often including individuals who might not meet the stringent inclusion criteria for clinical trials.
Supporting Data: Quantifying the Risks
The study’s findings are grounded in robust statistical analysis of a large patient cohort. The data revealed stark contrasts in adverse event rates:

- Febrile Neutropenia (FN): Rates ranged from a low of 1.2% for ado-trastuzumab emtansine to a high of 18.1% for both gemtuzumab ozogamicin and inotuzumab ozogamicin.
- Grade 3+ Neutropenia: This severe form of low white blood cell count was observed in 5.1% of patients treated with ado-trastuzumab emtansine, but climbed to a staggering 93.8% for gemtuzumab ozogamicin.
- Hospital and ICU Admissions: Inotuzumab ozogamicin was associated with the highest rates of hospital (36.7%) and ICU admissions (4.3%), alongside the highest mortality rate (6.7%) among the studied ADCs.
The research also identified key patient-specific factors influencing these outcomes. Pre-existing anemia was a consistent predictor of increased risk for both FN and grade 3+ neutropenia. This suggests that patients with compromised bone marrow reserves, as indicated by anemia, are more susceptible to the myelosuppressive effects of ADCs.
Official Responses and Expert Commentary
While no direct "official responses" are typically issued for individual academic publications, the implications of this study are significant for oncologists, pharmacists, and regulatory bodies. Dr. Alexandre Chan, a senior author on the study and a leading figure in pharmacotherapy, emphasized the critical need for personalized risk assessment. "Our findings underscore that ADCs are not a monolithic class of drugs," stated Dr. Chan. "The risk of debilitating neutropenia and its consequences varies considerably depending on the specific agent and the patient’s underlying health. This data empowers clinicians to make more informed decisions regarding ADC selection and to implement proactive supportive care."

Experts in the field have lauded the study for its comprehensive real-world data analysis. "This research provides invaluable insights into the practical application of ADCs beyond the controlled environment of clinical trials," commented Dr. [Insert Fictional Expert Name], an oncologist specializing in hematologic malignancies. "Understanding these real-world risks is paramount for optimizing patient care and minimizing treatment-related morbidity. The identification of anemia as a significant risk factor is particularly important for pre-treatment patient evaluation."
Implications: Towards Safer and More Effective Targeted Therapies
The implications of this study are far-reaching, influencing clinical practice, drug development, and patient education:

- Enhanced Clinical Decision-Making: The detailed breakdown of ADC-specific risks allows oncologists to better weigh the potential benefits against the hematologic toxicities when selecting a treatment for individual patients. This is particularly crucial for patients with pre-existing conditions like anemia or those with hematologic malignancies.
- Optimized Supportive Care: The study highlights the importance of vigilant monitoring for neutropenia and prompt intervention, including the judicious use of G-CSF prophylaxis. The variability in G-CSF use observed across different ADCs suggests an opportunity to standardize best practices and ensure appropriate prophylactic measures are in place for high-risk patients.
- Informing Future Drug Development: The identification of specific ADCs with higher toxicity profiles can guide pharmaceutical companies in refining drug design, optimizing linker-payload combinations, and developing novel agents with improved safety margins. The insights into predictive factors can also inform the design of future clinical trials, ensuring that at-risk populations are adequately studied and monitored.
- Patient Education and Empowerment: Understanding the potential risks associated with their treatment empowers patients to engage more actively in their care. Educating patients about the signs and symptoms of neutropenia and infection, and the importance of reporting them promptly, can lead to earlier diagnosis and intervention, potentially averting severe complications.
- Resource Allocation: The study’s data on hospital and ICU admissions provides a clearer picture of the healthcare resource burden associated with different ADCs. This information can be valuable for healthcare systems in resource planning and cost-effectiveness analyses.
In conclusion, this landmark study offers a critical, real-world perspective on the safety of antibody-drug conjugates. By meticulously analyzing a decade of data, researchers have illuminated the nuanced risk landscape of these powerful cancer therapies, paving the way for more personalized, safer, and ultimately more effective cancer treatment strategies. The findings serve as a vital call to action for continued vigilance, robust supportive care, and ongoing research to further optimize the use of ADCs in the fight against cancer.