Introduction: A Long-Overlooked Viral Threat
For over two decades, the medical community has grappled with a significant, yet frequently underappreciated, respiratory pathogen: the Human Metapneumovirus (HMPV). First isolated in 2001, HMPV has long been overshadowed by its more infamous relative, the Respiratory Syncytial Virus (RSV). However, recent comprehensive research—culminating in a 2026 review published in the journal Diagnostics—has finally pulled back the curtain on this virus, revealing it to be a formidable contributor to global respiratory morbidity.
While historically dismissed as a mild seasonal nuisance primarily affecting the immunocompromised or the elderly, contemporary data confirms that HMPV poses a severe risk to individuals across all age groups. With the capacity to cause everything from the common cold to life-threatening pneumonia and acute respiratory distress, HMPV is no longer just a "background" pathogen. It is a critical public health concern that demands urgent attention, improved diagnostic vigilance, and a dedicated strategy for vaccine development.

A Chronology of Discovery and Understanding
The history of HMPV is a classic case of a pathogen hiding in plain sight. While the virus was formally identified in 2001, retrospective serological investigations have traced its circulation among human populations for at least 70 years.
The Early Years (2001–2010)
Following its discovery, early research focused on the virus’s similarity to the Paramyxoviridae family, specifically its genetic kinship with avian pneumovirus and human RSV. Researchers quickly identified that while HMPV shared clinical features with RSV, its epidemiological behavior was distinct. During this decade, the focus remained largely on pediatric populations, as clinicians noted a high rate of infection among infants under five years of age.

Evolution and Global Spread (2011–2020)
As diagnostic capabilities improved, the global footprint of HMPV became clearer. Studies across Asia, Europe, and North America confirmed that the virus was present on every continent. Researchers noted that the virus displayed a distinct seasonal pattern, often peaking in late winter and early spring, frequently trailing behind the influenza and RSV seasons. This era also marked the beginning of more detailed genetic studies, identifying the A and B genotypes and their various sub-lineages, which began to show shifts in predominance across different geographic regions.
The Modern Era: Post-Pandemic Dynamics (2021–2026)
The COVID-19 pandemic significantly altered the circulation patterns of respiratory viruses. Strict public health measures and social distancing led to a temporary decline in HMPV transmission, particularly among school-aged children. However, as society returned to pre-pandemic norms, clinicians observed a resurgence of HMPV cases. The 2026 findings from the University of Tabuk and other international institutions have underscored that HMPV is not static; it is an evolving virus capable of shifting genotype predominance, which necessitates ongoing genomic surveillance.

Supporting Data: The Clinical and Molecular Burden
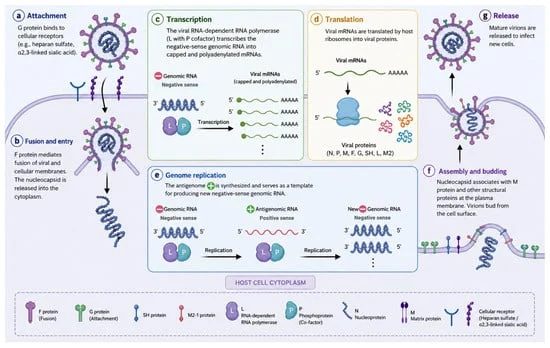
The molecular structure of HMPV is complex and elegant, featuring a negative-sense single-stranded RNA genome that encodes eight genes. Unlike RSV, HMPV lacks non-structural proteins (NS1 and NS2), which explains why the innate immune response to HMPV differs so significantly from its viral cousin.
Molecular Mechanisms of Disease
The HMPV genome’s reliance on the F (fusion) and G (attachment) glycoproteins is central to its pathogenicity. Research shows that the F protein is relatively stable, providing a target for potential vaccines, while the G protein exhibits high variability, likely an evolutionary adaptation to evade host immune defenses. The virus’s ability to interfere with mitochondrial antiviral signaling (MAVS) and inhibit the production of type I interferons is a hallmark of its immune-evasion strategy, effectively silencing the host’s early warning system and allowing the virus to establish a foothold in the respiratory epithelium.

Epidemiological Trends
Data from the 2026 Diagnostics review highlights the following:
- Prevalence: In some hospitalized populations, HMPV detection rates range between 4% and 12%, particularly during peak spring months.
- Severity: Approximately 11% of hospitalized patients with HMPV require ventilatory assistance, and 12% require intensive care unit (ICU) admission.
- The Adult Reality: Contrary to older beliefs, HMPV is a leading cause of severe pneumonia in the elderly, with mortality rates in some outbreaks reaching 11% in institutionalized care settings.
- Co-infection: HMPV is frequently found in co-infection with other pathogens, including RSV, influenza, and various bacteria like Streptococcus pneumoniae. While the exact impact of these co-infections on morbidity remains a subject of debate, the data suggests that they often complicate clinical management and prolong hospital stays.
Diagnostic Breakthroughs: From Culture to CRISPR
One of the primary reasons HMPV remained "neglected" for so long was the lack of efficient, routine diagnostic tools. Early methods relied on viral isolation in cell lines like Vero or LLC-MK2 cells—a process that was slow, labor-intensive, and lacked the sensitivity required for rapid clinical decision-making.

The current "gold standard" has shifted toward molecular diagnostics. Real-time RT-PCR (rRT-qPCR) has revolutionized the field, offering high sensitivity and specificity. However, the future of HMPV diagnostics is even more promising. Recent advancements include:
- Isothermal Amplification: Techniques like LAMP (Loop-mediated isothermal amplification) and RAA (Recombinase-aided amplification) offer the ability to detect the virus in under 30 minutes at consistent temperatures, eliminating the need for expensive thermal cycling equipment.
- CRISPR-Cas12a Integration: By combining isothermal amplification with CRISPR-based detection, clinicians can now achieve incredible sensitivity (detecting as few as 700 copies/mL) with results readable via simple lateral flow assays, akin to a pregnancy test.
- Metagenomic Sequencing (mNGS): This high-throughput technology allows researchers to unravel the entire viral genome, which is essential for monitoring how HMPV evolves and shifts its genotype, providing a blueprint for next-generation vaccines.
Implications for Public Health and Future Management
The recognition of HMPV as a major respiratory pathogen has profound implications for global healthcare policy.

The Call for Vaccination
Currently, there is no licensed vaccine for HMPV. The supportive care model—focusing on hydration, oxygen support, and antipyretics—is the only standard of care. However, the 2026 report highlights several promising avenues. Researchers are testing:
- Live Attenuated Vaccines (LAVs): By deleting specific genes (like SH or M2-2), scientists are creating strains that induce a robust immune response without causing disease.
- Chimeric Vaccines: Combining HMPV antigens with those from influenza or RSV has shown potential in preclinical models to provide "blanket" protection against multiple respiratory viruses.
- Subunit and VLP Vaccines: Virus-like particles (VLPs) that mimic the structure of the HMPV surface are proving effective in eliciting neutralizing antibodies in primate models.
Clinical Awareness
Healthcare providers must now include HMPV in their differential diagnoses for acute respiratory infections, particularly when testing for influenza and RSV returns negative. The persistence of viral shedding for up to two weeks after the onset of illness suggests that current isolation protocols in hospitals may need to be updated to prevent nosocomial (hospital-acquired) transmission.

Conclusion: A Shift in Perspective
The scientific consensus is clear: Human Metapneumovirus is a formidable public health challenge. It is no longer a "new" virus, nor is it a minor one. The evidence collected by researchers across the globe underscores the urgent need for a shift in strategy. From integrating advanced CRISPR-based diagnostic tools into routine clinical practice to accelerating the clinical trial pipeline for chimeric and subunit vaccines, the path forward is one of proactive engagement.
As we move further into the 2020s, the goal is to transform HMPV from a "frequently neglected" pathogen into a well-managed and preventable respiratory threat. The scientific community has provided the map; now, it is up to health authorities and policymakers to provide the resources to finish the job. Through increased surveillance and a commitment to innovation, we can mitigate the significant morbidity and mortality caused by this hidden, but no longer ignored, viral adversary.