In the complex landscape of pediatric medicine, few conditions present as many systemic challenges as Inflammatory Bowel Disease (IBD). Beyond the debilitating gastrointestinal symptoms of Crohn’s disease (CD) and ulcerative colitis (UC), young patients face a significantly elevated risk of life-threatening venous thromboembolism—a risk estimated to be six times higher than that of their healthy peers.
A recent study published in the journal Pharmaceuticals by a research team at the Medical University of Graz has unveiled a critical missing piece of this puzzle. By investigating the "hemostatic paradox"—the observation that active IBD patients exhibit both a prothrombotic state and a counterintuitive delay in the initiation of blood clotting—researchers have identified Tissue Factor Pathway Inhibitor (TFPI) as a key mediator in the disease’s inflammatory and coagulation crosstalk.
The Hemostatic Paradox: A Clinical Mystery
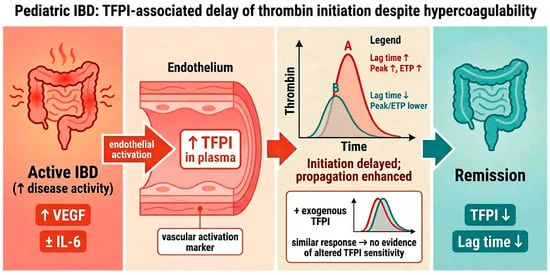
For years, clinicians have observed that children with active IBD show a unique coagulation profile. Using calibrated automated thrombography, researchers have consistently found that while these patients produce more thrombin overall—a hallmark of a hypercoagulable, or "clot-prone," state—the "lag time" before clotting begins is paradoxically prolonged.

"We have long observed that while the total potential for thrombin generation is high in active disease, the initial phase of the clotting process seems delayed," says lead researcher Axel Schlagenhauf. The research team hypothesized that this lag was not a sign of a "thinner" blood profile, but rather the result of an overabundance of natural anticoagulants released during the systemic inflammatory response, specifically TFPI.
Chronology of the Study
The research, conducted over a three-year period (2019–2022) at the Department of Pediatrics and Adolescent Medicine in Graz, involved a prospective longitudinal cohort of 25 children and adolescents (10 with Crohn’s disease and 15 with ulcerative colitis).
- Baseline Recruitment: Patients aged 7–18 were enrolled upon diagnosis, exhibiting classic symptoms such as hematochezia, abdominal pain, and diarrhea.
- Assessment Phases: Blood samples were meticulously collected at three intervals: at initial diagnosis (active disease), during early treatment (3–6 weeks post-medication), and upon achieving clinical remission (defined as low disease activity scores).
- Data Analysis: The team utilized advanced laboratory techniques, including chromogenic assays and ELISA, to measure TFPI levels, tissue factor activity (TFA), and inflammatory markers like VEGF and IL-6.
- Results Publication: Following extensive peer review, the findings were published on May 8, 2026, marking a significant advancement in understanding the link between vascular inflammation and coagulation.
Supporting Data: Connecting the Dots
The study’s data offers a compelling narrative of how inflammation rewires the body’s clotting system. The researchers found that TFPI levels were significantly elevated during the active phase of both CD and UC.

Crucially, the statistical analysis demonstrated a robust positive correlation between plasma TFPI levels and the duration of the thrombin generation lag time (r = 0.43, p < 0.05). When patients moved into remission, these levels normalized in tandem with the lag time.
"We saw a direct, longitudinal link," notes Dr. Schlagenhauf. "As the clinical disease activity scores—PCDAI for Crohn’s and PUCAI for Ulcerative Colitis—dropped, so too did the TFPI levels. This confirms that TFPI is not just a random bystander; it is a sensitive marker of the vascular inflammatory state associated with IBD."
While markers like IL-6 remained relatively stable in some patients, Vascular Endothelial Growth Factor (VEGF) showed a sharp decline upon remission, mirroring the resolution of vascular inflammation. The study also included a benchmark cohort of healthy young adults to ensure that the elevations observed were indeed pathological and not age-related physiological variances.

Official Perspectives and Expert Interpretation
The research team acknowledges that while the study provides a "hemostatic signature" for active IBD, it also poses new questions regarding treatment. One of the study’s most intriguing findings is that the reduction in TFPI and the corresponding normalization of lag time occurred consistently across different treatment regimens, including both biologic therapies (like anti-TNF agents) and non-biologic protocols.
"We initially wondered if specific drugs were driving this change," the authors noted in their discussion. "However, the uniformity of the shift across treatment strata suggests that the resolution of inflammation itself is the primary driver. The hemostatic system is essentially ‘resetting’ as the underlying disease activity is brought under control."
The authors emphasize that because TFPI acts as both an anticoagulant and a potential regulator of inflammation, it should be viewed as a multifunctional participant in IBD pathology. It may help suppress the matrix metalloproteinases that contribute to gut tissue destruction, making it a complex target for future therapeutic interventions.

Clinical Implications: A Path Toward Precision Medicine
The implications of this study are twofold: diagnostic and therapeutic.
1. Enhanced Risk Stratification
Standard clinical coagulation tests, such as PT (prothrombin time) and aPTT, are notoriously poor at capturing the subtle, high-stakes shifts in thrombin generation seen in IBD. The use of automated thrombography and the monitoring of TFPI levels could offer clinicians a more nuanced tool to assess the thrombotic risk of a patient. By identifying those with particularly high TFPI and prolonged lag times, doctors may be able to better tailor prophylactic care for patients at the highest risk of venous thromboembolism.
2. A New Therapeutic Target?
The dual role of TFPI—acting as an inhibitor of coagulation while potentially modulating the inflammatory environment—opens the door to new, targeted therapies. While the researchers caution that any manipulation of the coagulation cascade must be handled with extreme care to avoid bleeding risks, the study provides a roadmap for future research. If TFPI levels can be modulated to address the "vascular" side of IBD, it might provide a way to protect the intestinal barrier from inflammation-induced damage while simultaneously managing the patient’s systemic thrombotic risk.

Conclusion: Looking Toward the Future
This study reinforces the critical, often overlooked connection between the digestive system and the circulatory system. By proving that TFPI tracks with disease activity and explains the mysterious "lag time" in clotting, the researchers at the Medical University of Graz have provided a more granular understanding of pediatric IBD.
While the study’s sample size—25 patients—is reflective of the challenges inherent in pediatric clinical research, the findings are robust and provide a foundation for larger, multi-center trials. As the medical community moves toward more personalized care, markers like TFPI may soon become standard in the routine monitoring of pediatric IBD, helping to ensure that the next generation of patients not only achieve remission but do so with a significantly reduced risk of secondary complications.
Summary of Key Findings
- The Paradox: Active IBD patients show increased thrombin generation but a delayed "lag time" before clotting begins.
- The Culprit: Elevated Tissue Factor Pathway Inhibitor (TFPI) levels are responsible for this delayed lag time.
- The Link: TFPI levels correlate strongly with clinical disease activity and resolve upon treatment.
- Clinical Value: Monitoring TFPI could provide a more accurate way to assess thrombotic risk in pediatric IBD patients than standard blood tests.
- Next Steps: Future studies will investigate whether targeted modulation of TFPI could serve as a therapeutic strategy for managing both inflammation and hypercoagulability.