In the ongoing war against cancer, the human immune system remains our most potent weapon. Yet, for patients suffering from acute myeloid leukemia (AML), that weapon is often rendered ineffective. While modern immunotherapy—which has revolutionized the treatment of diseases like melanoma and lymphoma—has offered hope to many, AML has remained stubbornly resistant. A groundbreaking study published in the journal Science has now uncovered the culprit: a sophisticated, sugar-coated "cloak" that leukemia cells use to physically shield themselves from immune destruction.
Researchers from the Broad Institute of MIT and Harvard, Mass General Brigham, and the Dana-Farber Cancer Institute have identified a protein called CD43, which, when heavily glycosylated (coated in sugar molecules), creates a physical barrier that prevents immune cells from ever making the contact necessary to destroy the cancer. This discovery, which challenges long-held assumptions about how cancer cells signal their presence, could pave the way for a new generation of immunotherapies capable of dismantling this defensive armor.
The Main Facts: Decoding the "Don’t-Eat-Me" Misconception
For years, the scientific community has focused on a protein known as CD47, famously dubbed the "don’t-eat-me" signal. The prevailing theory held that cancer cells express high levels of CD47 to tell patrolling immune cells—specifically macrophages—that they are healthy and should not be consumed. This hypothesis led to the development of several experimental drugs designed to block CD47, intended to "de-cloak" the cancer cells.
However, clinical trials for these drugs in AML patients have largely yielded disappointing results. The new study provides a compelling explanation for these failures: CD47 may be a red herring.
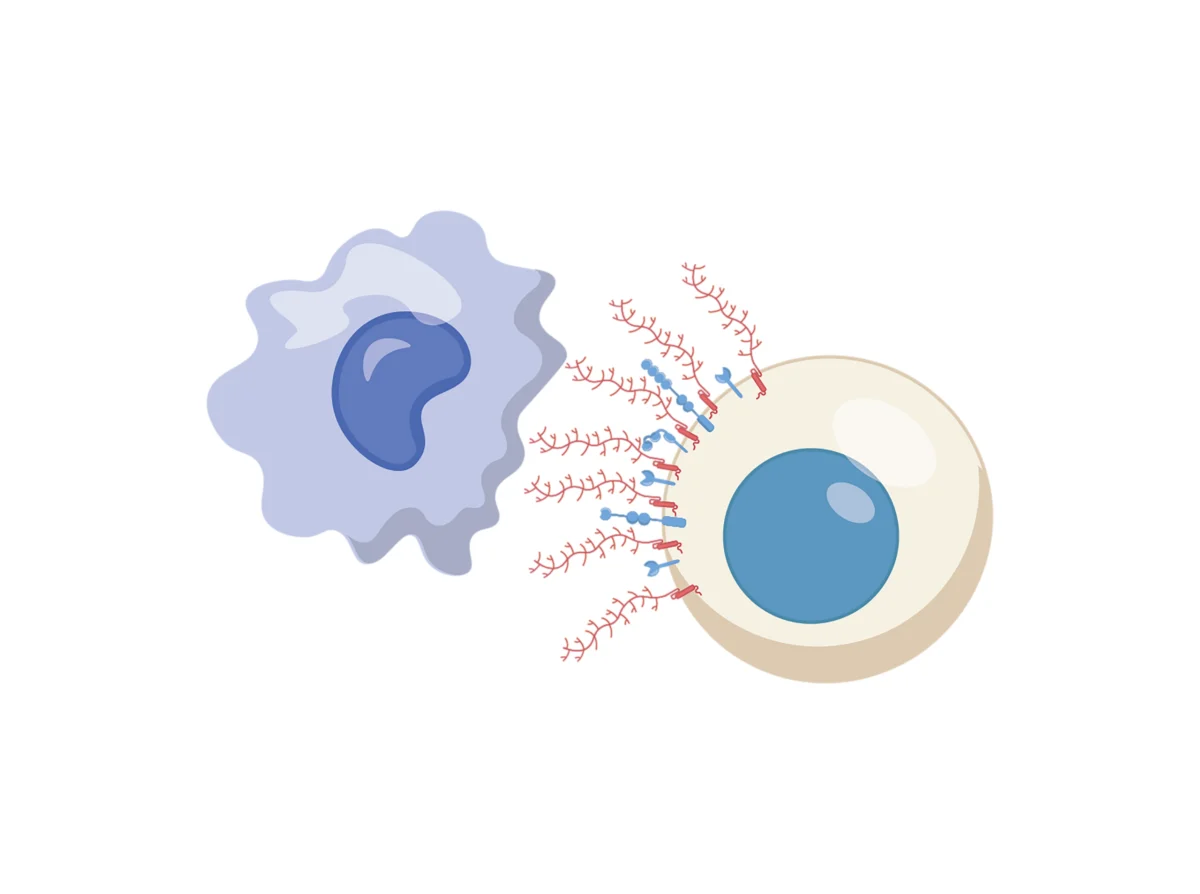
By conducting a genome-wide CRISPR screen, the researchers systematically disabled thousands of genes in millions of AML cells to determine which factors truly controlled immune interaction. To their astonishment, disabling CD47 had virtually no impact on whether macrophages could engulf the cancer cells. Instead, the team discovered that the true "shield" was CD43. When leukemia cells were stripped of CD43—or the specific enzymes responsible for attaching sugar molecules to it—the macrophages were suddenly able to recognize and consume the cancer cells with ease.
Chronology of a Scientific Breakthrough
The path to this discovery was born out of clinical frustration. Co-first authors Dr. Jooho Chung, a postdoctoral researcher in the Manguso lab and medical oncologist at Mass General Brigham, and Dr. Mounica Vallurupalli, a Dana-Farber medical oncologist and postdoctoral researcher in the Golub lab, frequently treated patients whose leukemia failed to respond to state-of-the-art immunotherapies.
"We know that leukemia can be susceptible to immune attack, but we also know that most cases of AML don’t respond to the newer immunotherapies that have worked so well in other cancers," Dr. Chung noted. "We wanted to understand why."
The Experimental Roadmap
- Genome-Wide CRISPR Screen: The team deployed CRISPR technology to disable genes across the entire AML genome. They then introduced macrophages to these modified cells to see which, if any, would be engulfed.
- The CD43 Discovery: The screen highlighted CD43 as the primary gatekeeper. Cells lacking CD43 were targeted by macrophages at significantly higher rates.
- Sugar Coating Analysis: Further testing revealed that the protection was not inherent to the protein itself, but to the sialic acid sugar coating.
- Physical Barrier Verification: The researchers created progressively shorter versions of the CD43 protein. They found that the shorter the protein, the more vulnerable the cell became to immune attack. This provided the "smoking gun" evidence that CD43 was acting as a physical, rather than a chemical, barrier.
- Cross-Immune Validation: The team expanded their testing to include T cells and Natural Killer (NK) cells. They found that inhibiting CD43 enhanced the efficacy of these immune cells as well, suggesting a universal mechanism of defense.
Supporting Data: Why Physical Proximity Matters
The study’s most profound realization is that the immune evasion mechanism is purely mechanical. In many cancers, signaling pathways are used to trick immune cells via chemical receptors (the ligand-receptor model). However, the evidence presented in Science suggests that CD43, when heavily glycosylated, acts like a dense forest of sugar trees that prevents immune cells from reaching the "cell membrane" surface.
When the researchers replaced CD43 with another sugar-coated protein, MUC1—which is found in various solid tumors—the same barrier effect was observed. This data suggests that the "sugar-shield" phenomenon is not unique to leukemia. Instead, it appears to be a foundational, perhaps even primitive, method of immune evasion that many cancer types may be exploiting to survive.
Dr. Robert Manguso, an associate member of the Broad Institute and co-leader of the study, reflected on the initial confusion during the validation phase: "We designed this experiment fully expecting that CD47 would be the most important protein for blocking the immune system. When it barely registered, we thought we’d made a mistake. We repeated the experiments over and over. But we kept getting the same answer."
Official Responses and Expert Perspectives
The academic community has received the findings with significant interest, as it redirects the focus of cancer immunotherapy research.
Todd Golub, director of the Broad Institute and a co-senior author of the study, emphasized the broader implications for the field: "Discovering this phenomenon opens up a new avenue for cancer immunotherapy. We think that the concept of cancers hiding behind sugar-coated proteins is not only important for leukemia, but likely other cancers as well."
The researchers are currently moving toward translational research, investigating antibodies that specifically bind to CD43. The goal is to develop a therapeutic agent that can effectively strip away this sugar-coated protection.
"If we could block CD43 in just the right way, that could go a long way to developing a new therapeutic strategy to kill these cancer cells and treat leukemia," said Dr. Mounica Vallurupalli. By neutralizing the physical barrier, clinicians hope to allow the immune system to do what it was designed to do: identify and eliminate malignant cells.
Implications: A New Horizon for Immunotherapy
The implications of this study are transformative. For decades, the focus of immunotherapy has been largely restricted to "releasing the brakes" on the immune system, such as using checkpoint inhibitors to prevent T-cell exhaustion. This research adds a new dimension: removing the physical obstacles that prevent immune cells from even "seeing" the target.
1. Re-evaluating Clinical Trials
The findings provide a clear rationale for why past clinical trials involving CD47 inhibitors may have failed. Future trials may need to pivot away from CD47 and toward targeting the glycan-coated CD43 proteins.
2. A Universal Mechanism of Defense
Because MUC1 and other glycoproteins are prevalent in solid tumors, the discovery of the "physical barrier" theory suggests that we may be looking at a common defense mechanism shared across oncology. If a therapy can be developed to "de-sugar" or block CD43 and similar proteins, it could potentially be adapted for breast, pancreatic, and lung cancers.
3. Precision Medicine
The ability to identify which patients express high levels of sugar-coated CD43 could allow for more precise patient selection in future clinical trials. By tailoring treatment to those whose cancers rely on this specific physical shield, doctors can increase the likelihood of success and minimize unnecessary side effects for patients who would not benefit from such a therapy.
Conclusion: Dismantling the Barrier
As research continues, the scientific team at the Broad Institute and their partners are working to turn these lab-based observations into clinical reality. The journey from identifying a protein’s function to developing a therapeutic antibody is complex, but the path is now much clearer.
By exposing the "sugar-coated" deception of leukemia cells, this study has not only provided a long-sought answer to why AML has remained elusive, but it has also provided a blueprint for dismantling the defenses of cancer cells everywhere. The "don’t-eat-me" era of cancer research may be fading, replaced by a new, more tactile understanding of how cancer hides in plain sight. For patients and clinicians alike, this represents a significant shift—one that turns a formerly impenetrable wall into a targetable vulnerability.