In a significant leap forward for computational oncology, researchers from Istanbul Topkapi University have unveiled a novel, contribution-aware AI framework designed to tackle the molecular complexity of lung adenocarcinoma (LUAD). The study, published in the May 2026 issue of the journal Pharmaceuticals, introduces a sophisticated metaheuristic approach to drug repurposing, offering a more nuanced strategy for selecting multi-drug combinations than traditional methods.
As cancer treatment shifts away from "one-size-fits-all" therapies, the ability to identify precise combinations of existing drugs—a process known as drug repurposing—has become a cornerstone of modern research. By focusing on the specific driver modules that fuel LUAD, this new computational model aims to provide clinicians and researchers with high-potential therapeutic hypotheses.
Main Facts: A New Frontier in Computational Oncology
Lung adenocarcinoma, a prevalent form of non-small cell lung cancer (NSCLC), is notoriously heterogeneous. Its molecular profile varies significantly between patients, rendering single-agent therapies frequently ineffective due to the rapid emergence of drug resistance. To counter this, scientists often turn to combination therapies; however, the sheer number of possible drug interactions makes experimental testing of every combination impossible.
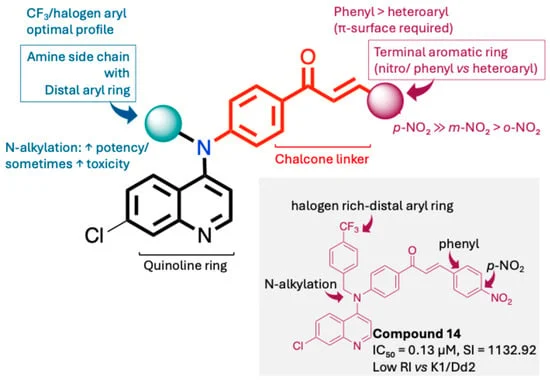
The research team, led by Sajjad Nematzadeh and Arzu Karaul of the Software Engineering Department at Istanbul Topkapi University, addressed this challenge by developing a "contribution-aware" framework. Unlike previous computational models that treat all dysregulated genes as equal, this system evaluates how specific drugs interact with specific gene targets.
Key Innovations of the Framework:
- Contribution-Aware Logic: The model utilizes a weighted contribution matrix derived from the Comparative Toxicogenomics Database, ensuring that the AI understands the direction and impact of drug-gene interactions.
- Genetic Algorithm Optimization: The framework employs a metaheuristic genetic algorithm to sift through massive datasets, identifying the most effective combinations of up to six drugs.
- Multi-Objective Fitness Function: The model does not just look for "reversal" of gene expression; it balances factors such as Mean Absolute Error (MAE), drug redundancy (waste), coverage of driver genes, and the overall complexity of the combination.
- Transcriptomic Validation: The results were cross-referenced with the CLUE (Connectivity Map) database to ensure that the suggested drug combinations effectively reverse the disease-state transcriptomic signature.
Chronology: The Development Path
The project followed a rigorous timeline of development, testing, and validation, culminating in its publication in the spring of 2026.
- April 7, 2026 – Initial Submission: The research team formally submitted their findings to Pharmaceuticals, having completed the design of the ten curated LUAD driver scenarios.
- April 2026 – Peer Review Phase: The study underwent intensive scrutiny, focusing on the robustness of the genetic algorithm and the biological relevance of the 44 unique genes targeted.
- May 4, 2026 – Revision Completion: The authors addressed feedback regarding the statistical significance of their findings and the limitations of their computational scope.
- May 6, 2026 – Acceptance: The manuscript was accepted, with reviewers highlighting the transparency of the scenario-based methodology.
- May 9, 2026 – Official Publication: The study was released in the journal’s 19th volume, providing the scientific community with an open-access resource for future cancer research.
Supporting Data: Why Small Combinations Work
One of the most compelling findings of the study concerns the "diminishing returns" of multi-drug combinations. The researchers tested combinations ranging from one to six drugs across ten different cancer scenarios.
The Power of Three
The data revealed that increasing the number of drugs from one to three yielded a dramatic improvement in the Mean Absolute Error (MAE)—the primary metric for how effectively a combination "reverses" the cancer gene signature. However, adding a fourth, fifth, or sixth drug provided only marginal gains in efficacy. This suggests that for most patients, a "triple therapy" approach—optimally selected via this AI framework—may provide the best balance between therapeutic efficacy and manageable complexity.
Performance Against Baseline
When compared to a standard baseline model that only considers MAE, the team’s contribution-aware objective outperformed or matched the baseline in 54 out of 60 test cases. This indicates that the inclusion of variables like "coverage" and "entropy" significantly enhances the model’s ability to predict drugs that work in harmony rather than in competition.
Cluster Identification
The AI identified distinct groups of interchangeable drugs. By mapping these, the researchers discovered shared mechanisms across different LUAD scenarios, suggesting that certain "repurposable" drugs could be utilized across a broader range of patient profiles than previously suspected.
Official Responses and Scientific Context
While the study has been met with enthusiasm, the authors are careful to maintain a professional, cautionary stance regarding its application.

"The proposed framework provides a transparent, scenario-based method for prioritizing repurposed drug combinations in LUAD," the authors stated in their conclusions. "The results are computational and hypothesis-generating. They should guide future experimental testing, not clinical treatment decisions."
This disclaimer is standard practice in bioinformatics, as computational predictions must always be validated in in vitro (cell culture) and in vivo (animal model) studies before clinical trials in humans can be considered. The research emphasizes that the goal is to accelerate the discovery phase, not to bypass the essential safety protocols of oncology.
Experts in the field have noted that the integration of the Comparative Toxicogenomics Database with genetic algorithms represents a significant step forward in transparency. By making the framework "contribution-aware," the researchers have made it easier for biologists to understand why the AI chose a specific drug, thereby increasing the "trust factor" of the model.
Implications: The Future of Precision Oncology
The implications of this study are far-reaching for both the pharmaceutical industry and clinical oncology.
1. Accelerating Drug Repurposing
Drug repurposing—the use of existing, approved drugs for new indications—is significantly faster and cheaper than traditional drug discovery. By using AI to identify combinations of existing drugs, researchers can theoretically bypass years of toxicity and safety testing, moving directly to validation of efficacy.
2. Addressing Tumor Heterogeneity
The "ten scenarios" approach used by Nematzadeh and Karaul is particularly significant. Because LUAD is so molecularly diverse, a single drug often fails because it only targets one pathway. By creating a model that can be tailored to different "scenarios" of gene dysregulation, the researchers have created a blueprint for personalized medicine that could eventually be applied to an individual patient’s biopsy data.
3. Reducing Treatment Costs
The inclusion of a "cost" parameter in the fitness function of the genetic algorithm suggests that the authors are thinking about the practicalities of healthcare delivery. By prioritizing cost-effective combinations, the framework could potentially lead to therapies that are not only more effective but also more accessible to a wider patient population.
4. A New Tool for the Research Pipeline
Moving forward, this framework serves as a "filter" for the scientific community. Instead of laboratory researchers testing thousands of random combinations, they can use the Istanbul Topkapi University model to narrow the list down to the top-tier candidates. This "AI-in-the-loop" approach is likely to become the standard in academic and commercial drug development labs over the next decade.
Closing Remarks
The work of Sajjad Nematzadeh and Arzu Karaul serves as a vital reminder of the potential of interdisciplinary research. By marrying the precision of genetic algorithms with the vast knowledge contained in toxicogenomics databases, they have provided a roadmap for tackling one of the most stubborn challenges in cancer care. As the medical community moves toward an era of data-driven, personalized interventions, frameworks like these will be essential in navigating the immense complexity of human disease.
For researchers and clinicians interested in applying this methodology, the full dataset and the logic behind the multi-objective fitness function are detailed in the published paper, providing a transparent foundation for the next generation of cancer therapy research.