London, UK – May 8, 2026 – In an era where clinical trial complexity is escalating, placing unprecedented demands on research sites, Paradigm Health has today announced the launch of its groundbreaking eSource Casebook. This innovative, EHR-integrated application aims to fundamentally transform data capture processes in clinical research, promising to significantly reduce the manual burden on site staff, accelerate trial timelines, and elevate data quality. The introduction of eSource Casebook arrives at a critical juncture, as the industry grapples with the inefficiencies and escalating costs associated with traditional, manual data entry methods.
The current landscape of clinical trials is characterized by increasingly intricate protocols, leading to a substantial increase in the volume and complexity of data that research sites must meticulously collect and manage. Studies indicate that manual data entry, a process historically fraught with time consumption and potential for error, has tripled in the past decade. This surge places immense strain on already stretched site resources, directly impacting their capacity to conduct research and, critically, delaying the generation of vital insights that drive medical advancements. On average, a staggering eight days can elapse between a patient visit and the subsequent data entry into electronic data capture (EDC) systems. This protracted lag not only slows the overall progress of a trial but also significantly inflates operational costs, impacting the economic viability of research endeavors.
While the concept of electronic source data (eSource) has long been recognized for its potential to alleviate these burdens, its widespread adoption and full benefit realization have been hampered by fragmented and siloed systems. These technological divisions prevent both pharmaceutical sponsors and research sites from achieving the seamless data flow and efficiency gains that eSource promises. To effectively address these systemic challenges, the clinical research industry has been in urgent need of purpose-built solutions. Such solutions must not only streamline the intricate process of data collection and reduce the workload on research personnel but also foster enhanced collaboration between all stakeholders, all without introducing additional operational overhead or causing further delays to already ambitious trial timelines. Paradigm Health’s eSource Casebook emerges as a direct response to this pressing industry demand.
Reducing the Data Capture Burden to Boost Efficiency and Research Capacity
The core innovation of eSource Casebook lies in its intelligent integration with Electronic Health Records (EHRs). This deep integration allows the application to proactively extract trial-relevant data from both structured and unstructured sources within the EHR. Unlike conventional methods that necessitate manual transcription, eSource Casebook automates the auto-population of electronic Case Report Forms (eCRFs). Furthermore, it facilitates the secure and efficient transmission of this captured data directly to sponsor Electronic Data Capture (EDC) systems.
Essentially, eSource Casebook acts as a sophisticated intermediary, taking over the site-facing responsibilities traditionally handled by sponsor EDC systems. By consolidating all source data extraction and initial entry into a single, intuitive application, it dramatically streamlines the data management process. This seamless integration is designed to fit harmoniously within the existing workflows of research teams, minimizing disruption and maximizing adoption.

Clinical Research Coordinators (CRCs) and other research staff can now enter data with unprecedented speed and accuracy. The application features an intuitive import workflow that guides users through the data selection process, complete with direct citations for quick and easy verification. This significant reduction in time spent on manual data entry and the alleviation of the overall data burden empowers research sites in several key ways. Firstly, it allows them to enhance the patient experience by freeing up staff time for direct patient interaction. Secondly, it significantly boosts their capacity to undertake a greater number of studies with the same or even fewer resources. Finally, it ensures a smoother and more efficient trial process for all participants, from the patient to the sponsor.
Efficient and Accurate Data Management: A Foundation for Reliable Research
The efficiency and accuracy of data management are paramount to the integrity and success of any clinical trial. eSource Casebook addresses this critical need through a multi-faceted approach:
- Automated Data Extraction: The system’s ability to connect directly to EHRs and intelligently extract relevant data points, both structured (e.g., lab results, vital signs) and unstructured (e.g., physician notes, imaging reports), drastically reduces the need for manual transcription. This automation minimizes the risk of human error inherent in manual data entry.
- Intelligent Data Parsing: Utilizing advanced algorithms, including Natural Language Processing (NLP) and Large Language Models (LLMs), eSource Casebook can interpret and extract crucial information from unstructured text within EHRs. This capability unlocks a wealth of previously difficult-to-access data, enriching the trial dataset without requiring extensive manual review.
- eCRF Auto-Population: Once data is extracted and parsed, eSource Casebook automatically populates the relevant fields within the eCRF. This feature ensures consistency and adherence to protocol requirements, as the data is directly mapped from its source to the designated fields.
- Real-time Data Validation: The application can incorporate built-in validation rules, flagging potential discrepancies or missing information at the point of entry. This proactive approach to data quality ensures that errors are identified and corrected early in the process, preventing downstream issues and the need for costly data reconciliation.
- Streamlined Source Data Verification (SDV): By providing a clear, integrated view of both the source data and the populated eCRFs, eSource Casebook significantly simplifies the SDV process. Auditors and monitors can more easily compare the data entered into the EDC with the original source documents, enhancing the efficiency and effectiveness of site audits.
Empowered and Productive Research Teams: Driving Site Performance
The impact of eSource Casebook extends beyond mere data management; it directly empowers research teams, fostering a more productive and positive work environment:
- Reduced Cognitive Load: By automating repetitive and time-consuming tasks, eSource Casebook liberates research coordinators and investigators from the mental strain of manual data entry and cross-referencing. This allows them to focus on higher-value activities, such as patient care and scientific interpretation.
- Enhanced Workflow Integration: The application is designed to seamlessly integrate into existing research workflows, minimizing the need for extensive retraining or the adoption of entirely new, complex systems. This user-centric approach promotes faster adoption and greater user satisfaction.
- Increased Time for Patient Engagement: With less time dedicated to administrative tasks, research staff can devote more attention to patient interaction, recruitment, and retention. This improved patient engagement can lead to better adherence to study protocols and a more positive overall trial experience for participants.
- Improved Job Satisfaction: The reduction of tedious manual tasks and the increase in efficiency can lead to greater job satisfaction among research site staff. This, in turn, can contribute to lower staff turnover and a more stable, experienced research workforce.
- Capacity for More Research: By optimizing operational efficiency, research sites can significantly increase their capacity to participate in multiple clinical trials simultaneously. This not only benefits the individual sites but also contributes to the overall acceleration of drug development by increasing the number of available research opportunities.
Scalable and Dynamic Infrastructure: Future-Proofing Clinical Research
In a rapidly evolving technological landscape, the infrastructure supporting clinical research must be both scalable and adaptable. eSource Casebook is built with these principles in mind:
- Cloud-Native Architecture: The application leverages a modern, cloud-native architecture, ensuring scalability to accommodate trials of any size, from small, early-phase studies to large, multi-site global trials. This flexibility allows the system to grow and adapt as the needs of research projects evolve.
- Interoperability Standards: eSource Casebook adheres to industry-recognized interoperability standards, such as FHIR (Fast Healthcare Interoperability Resources). This commitment to open standards ensures seamless integration with a wide range of EHR systems and other healthcare IT platforms, facilitating broader adoption and data exchange.
- Continuous Improvement: Paradigm Health is committed to the ongoing development and enhancement of eSource Casebook. Regular updates and feature enhancements will ensure that the application remains at the forefront of technological innovation, incorporating new advancements in AI, data analytics, and regulatory compliance.
- Robust Security and Compliance: Recognizing the sensitive nature of patient data, eSource Casebook is built with robust security measures and adheres to stringent regulatory compliance requirements, including HIPAA, GDPR, and other relevant data privacy regulations. This ensures the integrity and confidentiality of all captured data.
- Adaptability to Evolving Protocols: As clinical trial protocols become more sophisticated, eSource Casebook’s flexible design allows it to adapt to new data requirements and capture methodologies. This inherent adaptability ensures its continued relevance and utility in the face of future research challenges.
Connecting to the EHR and Integrating into Existing Workflows: A Seamless Transition
The true power of eSource Casebook lies in its ability to bridge the gap between clinical practice and clinical research data management. By establishing direct connections with EHR systems, the application eliminates the need for disparate data entry points and manual data reconciliation.

Direct EHR Data Import via FHIR: The system utilizes the FHIR standard to directly import structured data from EHRs. This ensures that essential patient information, such as laboratory results, medication lists, vital signs, and diagnoses, is accurately and efficiently transferred. This standardized approach minimizes the risk of misinterpretation and ensures data consistency.
Leveraging LLMs for Unstructured Data Extraction: A significant hurdle in EHR integration has been the challenge of extracting meaningful data from unstructured sources like physician notes, operative reports, and radiology findings. eSource Casebook employs advanced Large Language Models (LLMs) to intelligently parse this free-text information, identifying and extracting key clinical details. This capability unlocks a wealth of previously underutilized data, providing a more comprehensive picture of the patient’s health status and study participation.
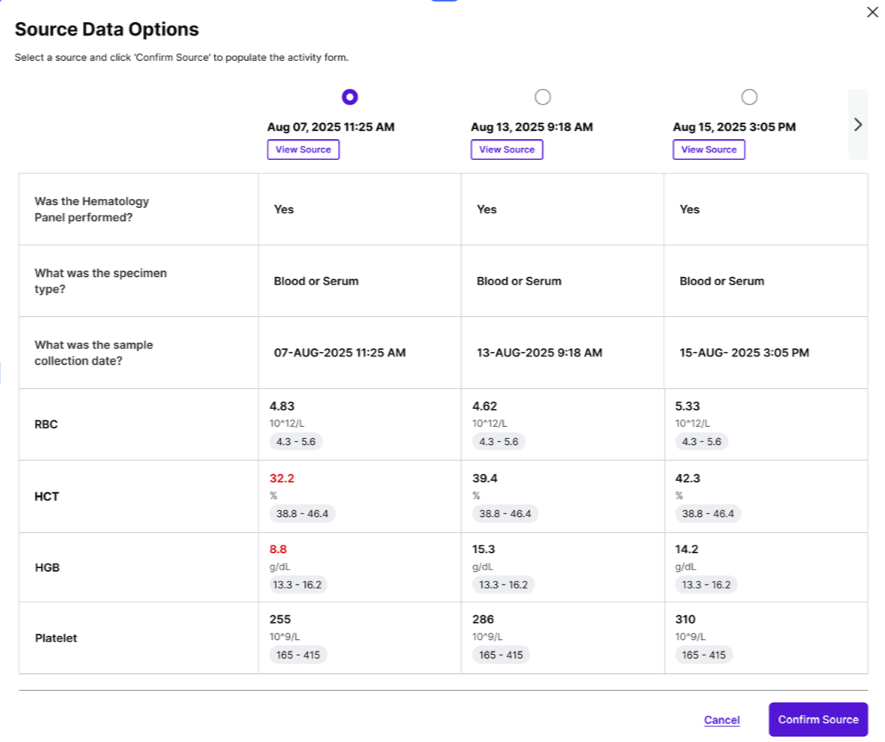
Streamlined Source Selection and Accurate eCRF Entry: The application provides users with quick, EHR-like views of pre-filtered data. This intuitive interface allows research staff to easily select the precise data required for the eCRF without the need to navigate multiple disparate systems. This significantly reduces the mental overhead and ensures that accurate data is consistently entered into the electronic case report forms.
Review of Auto-Populated eCRFs: Once data has been extracted and processed, eSource Casebook presents auto-populated eCRFs for review. The EHR data is intelligently translated into the expected format for the EDC, minimizing the need for manual adjustments or reformatting. This feature further reduces the burden on site staff and enables a smoother, more efficient submission process to the sponsor EDC.
The accompanying visual aids within the original announcement—demonstrating the intuitive interface for data review and the clear presentation of auto-populated eCRFs—underscore the user-centric design philosophy behind eSource Casebook. These features are critical for ensuring rapid adoption and maximizing the benefits of the system.
Implications for the Future of Clinical Trials
The introduction of Paradigm Health’s eSource Casebook marks a significant step forward in the evolution of clinical trial operations. By addressing the critical pain points of manual data entry, system fragmentation, and site resource strain, the application has the potential to:
- Accelerate Drug Development: By reducing the time it takes to collect, verify, and submit data, eSource Casebook can significantly shorten overall trial timelines, bringing life-saving therapies to patients faster.
- Reduce Operational Costs: The automation of data capture and the reduction in manual effort translate directly into cost savings for both research sites and sponsors, making clinical research more economically sustainable.
- Enhance Data Quality and Integrity: Minimizing manual transcription and leveraging automated validation processes leads to more accurate and reliable data, which is crucial for robust scientific analysis and regulatory submissions.
- Improve Site Sustainability: By making research more efficient and less burdensome, eSource Casebook can help to sustain and grow the network of research sites, ensuring continued capacity for clinical research.
- Democratize Access to Research: Increased efficiency and reduced costs could potentially lead to more research being conducted, offering more opportunities for patients to participate in clinical trials.
A Call to Action
Paradigm Health is inviting the clinical research community to embrace this transformative technology. The company emphasizes that by accelerating data entry, reducing auditing costs, and ensuring faster, more accurate data flow, eSource Casebook offers a compelling solution to long-standing industry challenges.
For organizations interested in learning more about how eSource Casebook can revolutionize their clinical trial operations, Paradigm Health encourages them to visit their website at paradigm.inc/solutions/conduct-esource-casebook or contact their team directly. The future of efficient, accurate, and accelerated clinical research is here, and it is powered by intelligent data integration.