
By Medical Editorial Staff | September 03, 2025
Receiving a breast cancer diagnosis is a life-altering event that thrusts patients into a whirlwind of complex medical terminology, urgent decision-making, and emotional turbulence. In the era of precision medicine, the standard of care has shifted away from a "one-size-fits-all" approach toward highly individualized treatment plans. Central to this evolution are two distinct yet frequently confused diagnostic tools: genetic testing and biomarker (genomic) testing.
While both rely on the study of genes to inform clinical decisions, they serve entirely different masters. One looks backward to the blueprint of your life, while the other looks at the immediate, erratic behavior of the tumor itself. Understanding this difference is not just an academic exercise; it is an essential step for any patient aiming to advocate for the most effective, personalized care possible.
Main Facts: The Core Divergence
To demystify these tests, it is helpful to establish their primary roles.
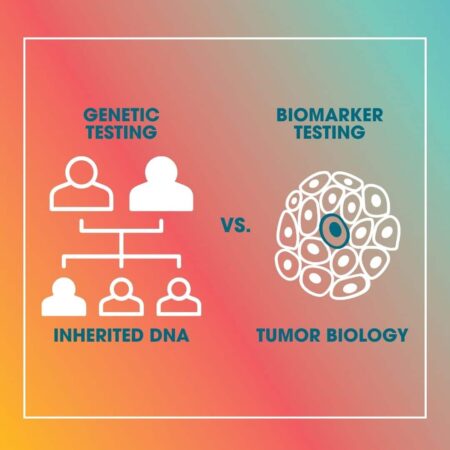
Genetic Testing is focused on the germline—the DNA you inherited from your biological parents. It is a diagnostic tool used to identify hereditary mutations (such as BRCA1 or BRCA2) that predispose individuals to cancer. Its primary value lies in risk assessment, surgical planning (such as preventative mastectomies), and informing family members of their own potential health risks.
Biomarker Testing—often referred to as genomic testing—is focused on somatic mutations. It is performed on a biopsy sample of the actual tumor. It ignores your inherent DNA to focus solely on the "personality" of the cancer cells. By analyzing the expression levels of specific genes within the tumor, oncologists can determine how aggressive the cancer is, how likely it is to recur, and, crucially, whether the patient will derive clinical benefit from systemic therapies like chemotherapy.
Chronology: When and Why These Tests Are Ordered
The timeline for these tests is distinct, reflecting their unique purposes in the clinical pathway.

The Genetic Testing Window
Genetic testing is typically considered at the point of diagnosis or even before, depending on family history. If a patient presents with a strong history of breast or ovarian cancer, genetic counseling is often the first step. The results here guide immediate surgical decisions—for example, a patient who discovers they carry a high-risk mutation may opt for a bilateral mastectomy rather than a lumpectomy, as the genetic finding implies a higher risk of a second, independent primary cancer.
The Biomarker Testing Window
Biomarker testing is almost exclusively performed post-diagnosis. Once a pathologist has confirmed a Stage I or II invasive breast cancer, the oncologist must determine the optimal adjuvant treatment. This is the moment for genomic assays like MammaPrint or BluePrint. These tests provide a "recurrence score" or a molecular subtype classification that helps the care team decide if the potential toxicity of chemotherapy is justified by the biological makeup of the tumor.
Supporting Data: Why Precision Matters
The medical community has moved away from relying solely on clinical features—such as tumor size, lymph node involvement, and grade—to determine treatment. While these factors are important, they are often insufficient to predict how a tumor will respond to specific drugs.
Recent clinical data emphasizes the following:
- De-escalation of Care: In many cases, biomarker testing reveals that a tumor has a low risk of recurrence despite its size or grade. This allows patients to avoid the debilitating side effects of chemotherapy, opting instead for endocrine therapy alone.
- Subtype Identification: BluePrint testing, for example, can classify a tumor into functional molecular subtypes (Luminal A, Luminal B, HER2-type, or Basal-type). This classification often aligns better with biological behavior than traditional immunohistochemistry (IHC) testing, ensuring the patient receives the therapy most likely to target the tumor’s specific vulnerabilities.
- Hereditary Insights: Genetic testing identifies that roughly 5–10% of all breast cancer cases are hereditary. For these patients, the identification of a mutation can be lifesaving not just for them, but for their children, siblings, and extended family members who may now qualify for early screening programs.
Official Responses: Clinical Perspectives
Leading oncological organizations, including the American Society of Clinical Oncology (ASCO) and the National Comprehensive Cancer Network (NCCN), have updated their guidelines to integrate these tests.
Experts emphasize that the "stronger" treatment is not always the "smarter" treatment. The clinical consensus is that biomarker testing is a critical tool to prevent over-treatment. By accurately identifying which tumors are truly indolent (slow-growing) versus those that are aggressive, clinicians can protect the patient’s quality of life without compromising survival rates.
Regarding genetic testing, the clinical response is centered on "cascade testing." Once an index patient (the person with cancer) is identified with a pathogenic mutation, their family members can undergo targeted testing. This proactive approach transforms the diagnosis from a solitary burden into an opportunity for familial health management.
Implications: Empowering the Patient
The shift toward personalized medicine places a new level of responsibility on the patient. To navigate this landscape effectively, patients should be prepared to discuss these options during their initial consultations.
Essential Questions for Your Care Team:
- Regarding Genetic Testing: "Given my personal and family history, should I meet with a genetic counselor to discuss germline testing?"
- Regarding Biomarker Testing: "Is my tumor eligible for genomic assay testing (such as MammaPrint)? How will the results of this test change my treatment plan?"
- Regarding Treatment Strategy: "If the genomic test shows a low risk of recurrence, does that mean I can safely omit chemotherapy?"
A Summary of Differences
| Feature | Genetic Testing | Biomarker Testing |
|---|---|---|
| Focus | Inherited DNA (Germline) | Tumor DNA/RNA (Somatic) |
| Objective | Assess hereditary cancer risk | Assess tumor biology/behavior |
| Primary Goal | Prevention and surgical planning | Treatment optimization (chemo decisions) |
| Scope | Benefits patient and family | Benefits the patient’s immediate treatment |
Conclusion: The Path Toward Smarter Care
The distinction between these two forms of testing is foundational to modern oncology. Genetic testing provides a map of your inherited vulnerabilities, helping you and your family manage future risks. Biomarker testing provides a roadmap for the current battle, allowing your doctor to tailor chemotherapy and other treatments to the specific biological signature of your cancer.
By understanding these tools, you are better equipped to engage in a dialogue with your oncology team. Remember that the ultimate goal of modern cancer care is to achieve the best possible outcome with the most precise intervention. When your treatment plan is based on your unique biology rather than broad clinical averages, you are receiving care that is, by definition, smarter.
As you navigate this journey, lean on your care team to explain the nuances of your specific reports. Advocate for your right to understand the "why" behind every recommendation, and never hesitate to ask for clarity. Knowledge is the most effective tool in your arsenal, turning complex medical data into a clear path toward healing.
Disclaimer: The information provided in this article is for educational purposes only and does not constitute medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.







