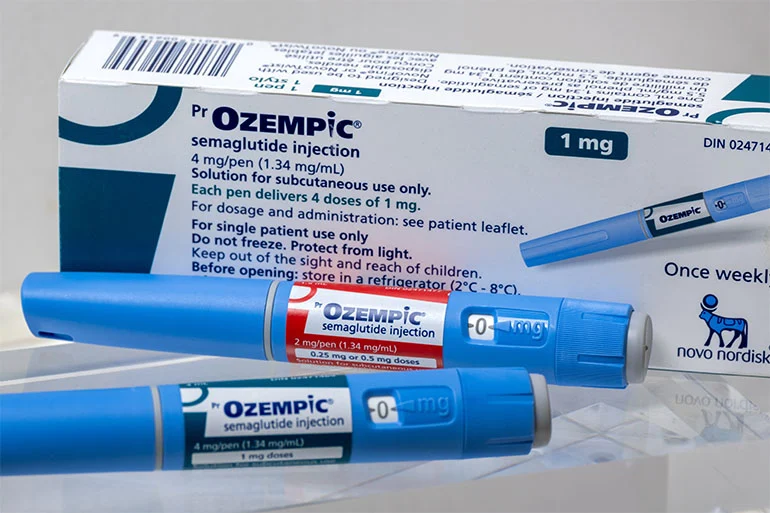
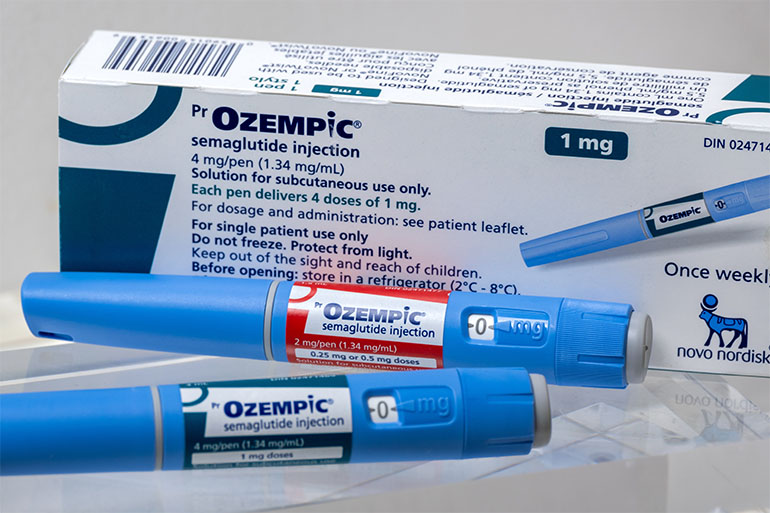
In a historic development that marks a watershed moment for the global pharmaceutical industry, Canada has become the first G7 nation to authorize a generic version of semaglutide. This breakthrough, led by the approval of Dr. Reddy’s Laboratories’ generic injection, represents a dramatic departure from the status quo that has defined the GLP-1 (glucagon-like peptide-1) receptor agonist market since the explosive rise of Novo Nordisk’s Ozempic.
As of April 2026, the Canadian regulatory landscape has shifted, effectively ending the monopolistic grip that high-priced, brand-name GLP-1 therapies have held over the metabolic disease sector. With over one million Canadians currently reliant on semaglutide for the management of type 2 diabetes and obesity, this regulatory green light signals the beginning of a new era of access, affordability, and competition.
Main Facts: The End of a Monopoly
The authorization of Dr. Reddy’s generic semaglutide—followed shortly by an approval for Apotex on May 1, 2026—is not merely a win for Canadian patients; it is a global case study in the vulnerability of patent-protected blockbuster drugs.
Health Canada’s assessment concluded that these generic versions are pharmaceutically equivalent to the reference product, Ozempic, meeting the highest standards for safety, efficacy, and quality. The approvals cover 2 mg/pen and 4 mg/pen presentations at a concentration of 1.34 mg/mL. For Dr. Reddy’s, the achievement highlights their mastery of complex peptide manufacturing, with the company confirming that the active pharmaceutical ingredient (API) is produced entirely in-house, with finished product manufacturing managed by OneSource Specialty Pharma.
The primary driver of this development is an administrative oversight that has become the stuff of industry legend: in 2019, Novo Nordisk failed to pay a relatively nominal $250 CAD maintenance fee for Canadian patent CA 2601784. While the company later characterized the lapse as a "strategic choice," the result was the permanent forfeiture of patent protection in the Canadian market.
Chronology of a Regulatory Turning Point
The path to this moment is paved with years of patent management, regulatory scrutiny, and a unique administrative blunder.
- October 2018: Novo Nordisk makes its final maintenance-fee payment on patent CA 2601784, "Acylated GLP-1 compounds."
- 2019: The critical $250 CAD fee is missed. The patent, which would have offered protection until March 2026 (and potentially through 2028 via a certificate of supplementary protection), begins to lapse.
- August 2020: The window for the reversal of the patent lapse officially closes. The patent is classified by the Canadian Intellectual Property Office as "Expired and beyond the Period of Reversal."
- 2022–2025: A period of intense global shortage for semaglutide begins. In the U.S., the FDA permits compounding pharmacies to fill the supply gap, a practice that formally ends in early 2025 as the supply chain stabilizes.
- April 2026: Health Canada approves Dr. Reddy’s generic semaglutide, officially becoming the first G7 regulator to permit competition in the segment.
- May 2026: Apotex receives its own approval for generic semaglutide, confirming that the Canadian market is now open for broader generic entry. Currently, seven additional submissions are under review.
Supporting Data: The Scale of the Semaglutide Phenomenon
To understand the magnitude of this shift, one must look at the financial dominance of the molecule. Semaglutide has evolved into perhaps the most commercially successful molecular franchise in modern pharmaceutical history.
In fiscal year 2025 alone, Novo Nordisk reported total sales for its GLP-1 portfolio—Ozempic, Wegovy, and Rybelsus—exceeding DKK 228 billion. Ozempic alone accounted for DKK 127.1 billion, cementing its status as the best-selling drug in Canada, where it generated $2.9 billion in revenue in 2025—a figure more than triple that of its closest competitor.
The "GLP-1 revolution" has fundamentally changed the metabolic disease landscape, setting a bar for commercial scale that has forced competitors, most notably Eli Lilly with its dual GIP/GLP-1 agonist tirzepatide, to accelerate their own pipeline developments. However, while the Canadian market has opened, the situation remains vastly different in the U.S., Europe, and Japan. In these regions, Novo Nordisk holds active patent protection on the semaglutide molecule until at least 2031 or 2032.
Official Responses and Strategic Implications
The fallout from the Canadian patent expiry has triggered a flurry of legal and corporate activity. Novo Nordisk has been proactive in the U.S. market, navigating a series of Paragraph IV patent challenges from a coalition of generic manufacturers, including Alvogen, Sun Pharma, and Mylan. While the company has settled many of these U.S.-based disputes, the terms remain strictly confidential, highlighting the company’s intent to protect its remaining global exclusivity at all costs.
Health Canada, for its part, has maintained a rigorous focus on the clinical integrity of the generic products. By classifying generic semaglutide as a "complex synthetic product," the agency has signaled that it will not compromise on the stringent requirements needed to demonstrate bioequivalence for injectable peptides.
Dr. Reddy’s Laboratories has framed the Canadian approval as a cornerstone of its "complex generics" strategy. By controlling the supply chain from the peptide API to the final injection pen, they are insulating themselves from the volatility that has historically plagued the GLP-1 market.
Implications: The Future of Access and Pricing
The arrival of generic semaglutide in Canada carries profound implications for the global pharmaceutical industry.
1. The End of the "Shortage Era"
For years, the global supply of semaglutide has been hampered by manufacturing bottlenecks, leading to severe shortages. The entry of generic manufacturers, who have now secured regulatory approval, suggests that production capacity will significantly expand. As more players enter the Canadian market, the pressure on the existing supply chain will likely ease, potentially lowering costs for provincial health plans and private insurers.
2. A New Blueprint for Patent Management
The $250 CAD administrative failure by Novo Nordisk will be studied by corporate legal teams for decades. It serves as a stark reminder that in the high-stakes world of blockbuster drugs, the smallest administrative detail can render a multi-billion-dollar patent portfolio obsolete. This incident has already caused other pharmaceutical firms to audit their international intellectual property filings with renewed vigor.
3. The Widening Gap Between Markets
The disparity between Canada and the rest of the G7 creates a "dual-track" global market. Canadian patients and healthcare systems will likely benefit from lower prices years ahead of their counterparts in the U.S. or Europe. This divergence may spark renewed political debates in other nations regarding the balance between incentivizing innovation and ensuring patient access to life-saving, off-patent medications.
4. The Rise of "Complex" Generics
Semaglutide is a complex peptide, and its successful genericization proves that the pharmaceutical industry has matured in its ability to replicate sophisticated biological or synthetic molecules. This shift will likely encourage more investment in the development of generic versions of other complex, high-value drugs, potentially disrupting the long-held assumption that biologics and complex peptides are "un-genericizable."
Conclusion
Canada’s approval of generic semaglutide is a historic marker of change. While the circumstances—a lapsed patent fee—were singular, the outcome represents a permanent shift in the commercial trajectory of the most important drug franchise of the 21st century. As the market in Canada moves toward full competition, the rest of the world will be watching closely to see how the influx of generic alternatives influences prices, clinical access, and the broader, global battle for dominance in the metabolic disease space. The era of the semaglutide monopoly, at least in the North, has officially drawn to a close.