In a major development for global ophthalmology, a comprehensive, multicenter study has provided robust real-world evidence confirming that the biosimilar ranibizumab, Ranieyes, is clinically comparable to the innovator drug, Lucentis (Accentrix). Published in the journal Pharmaceuticals (2026), the study, titled "Real-World Comparison of Biosimilar Ranibizumab (Ranieyes) and Innovator Ranibizumab (Lucentis/Accentrix) Across Multiple Retinal Vascular Diseases (The BRIO Study)," offers a compelling argument for the expanded use of cost-effective alternatives in the treatment of sight-threatening conditions.
The study analyzed nearly 5,000 eyes across five tertiary eye-care centers in India, representing one of the largest real-world assessments of its kind. By demonstrating that biosimilars can achieve similar visual and anatomical outcomes to their more expensive counterparts, the findings could signal a paradigm shift in how healthcare systems manage chronic retinal diseases, particularly in low- and middle-income regions.
1. Main Facts: Bridging the Gap in Ophthalmic Care
Retinal vascular diseases—including neovascular age-related macular degeneration (nAMD), diabetic macular edema (DME), retinal vein occlusion (RVO), and myopic choroidal neovascularization (mCNV)—are among the leading causes of vision loss worldwide. These conditions share a common reliance on intravitreal anti-vascular endothelial growth factor (anti-VEGF) injections to stabilize or improve vision.
However, the "treatment burden"—the requirement for frequent, often lifelong injections—presents a dual challenge: clinical exhaustion and severe financial strain. For many patients, the prohibitive cost of innovator anti-VEGF therapies leads to treatment non-adherence, ultimately resulting in permanent vision loss.

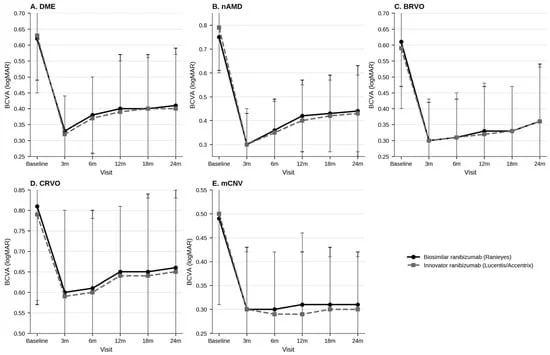
The BRIO study evaluated 4,997 eyes from 3,577 patients, comparing the performance of the biosimilar Ranieyes against the innovator drug. The findings were clear: across every major disease category, the biosimilar matched the performance of the innovator, showing equivalent improvements in visual acuity and central subfield thickness (CST) without any new or unexpected safety signals.
2. Chronology of the Research
The retrospective study spanned a rigorous assessment period, covering clinical data collected from July 2022 to October 2025.
- Initial Phase (2022): Researchers at five leading Indian eye-care institutions—including Disha Eye Hospitals and Nethralayam Superspeciality Eye Hospital—began the retrospective review of patient medical records.
- Data Aggregation (2023–2024): The team compiled data from over 21,000 total injections, ensuring a diverse and statistically significant sample size covering various retinal pathologies.
- Analysis and Review (Early 2026): After conducting longitudinal analyses of visual outcomes and safety profiles, the authors finalized their findings in March 2026.
- Peer Review and Publication (April–May 2026): Following a rigorous peer-review process, the study was accepted on April 26, 2026, and officially published on May 11, 2026.
3. Supporting Data: Evidence of Comparability
The strength of the BRIO study lies in its granularity. By tracking patients over a 24-month period, the researchers were able to observe not just the immediate efficacy of the drugs, but their long-term stability.
Visual and Anatomical Outcomes
Both the biosimilar and innovator groups demonstrated early improvements in Best-Corrected Visual Acuity (BCVA) and reductions in retinal fluid (CST). In cases of DME and RVO, the visual trajectories of the two groups were virtually indistinguishable. While a minor, isolated difference in nAMD outcomes was noted at the 12-month mark, it was clinically insignificant and failed to persist through the 18- and 24-month follow-up visits.

Safety and Tolerability
One of the most critical concerns for clinicians when switching to a biosimilar is safety. The study found that serious ocular adverse events, such as endophthalmitis, were extremely rare in both cohorts (occurring at a rate of 0.09/1000 for the biosimilar vs. 0.20/1000 for the innovator). Systemic vascular events were similarly infrequent, confirming that Ranieyes does not pose a heightened risk profile compared to the standard innovator treatment.
Injection Burden
The study also evaluated the frequency of injections required to manage these conditions. Because the injection burden was found to be comparable, researchers concluded that the positive outcomes were not the result of one group receiving significantly more or less treatment than the other, but rather a direct reflection of the molecule’s efficacy.
4. Official Interpretations and Clinical Context
The authors of the study, led by Dr. Debdulal Chakraborty, have emphasized that while the data is highly encouraging, it should be viewed as "observational comparative evidence" rather than a formal, randomized equivalence trial.
In their discussion, the researchers addressed the socioeconomic impact of the study. At the participating centers, the biosimilar Ranieyes was priced at approximately INR 12,000–16,000 ($129–$173), nearly half the cost of the innovator drug (INR 25,000–30,000 / $270–$325). This pricing disparity is the cornerstone of the study’s argument: by utilizing biosimilars, healthcare systems can drastically reduce the barrier to entry for patients, allowing more individuals to receive timely and continuous care.

"In settings where out-of-pocket expenditure remains an important determinant of treatment uptake and continuity, evidence supporting comparable real-world outcomes with a biosimilar agent may improve access to anti-VEGF therapy without materially compromising effectiveness or safety," the authors noted in their concluding remarks.
5. Implications for Global Health
The implications of the BRIO study extend far beyond the clinics of India. As the global population ages and the prevalence of diabetic eye disease continues to climb, the sustainability of current treatment models is under threat.
Enhanced Accessibility
The study provides a roadmap for other nations and healthcare providers to confidently incorporate biosimilars into their treatment protocols. By demonstrating that there is no "clinical penalty" for choosing a lower-cost option, the researchers provide physicians with the clinical confidence required to prioritize patient access.
Future Research Directions
The authors noted that their study was limited to a single brand of ranibizumab biosimilar. They explicitly called for further multicenter comparative studies across different biosimilar brands and more diverse patient populations. Additionally, while the study provides strong evidence for clinical equivalence, the authors encouraged future research to focus on formal cost-effectiveness modeling to quantify the total economic benefit of shifting to biosimilars.

A New Standard for "Real-World" Evidence
By utilizing a large, multi-institutional dataset, the BRIO study sets a high bar for real-world evidence (RWE) in ophthalmology. It highlights the importance of transparency in reporting—including detailed data on adverse events and longitudinal visual trends—which is essential for building physician trust in biological alternatives.
As the medical community moves toward a more value-based approach to care, the findings regarding Ranieyes serve as a landmark study. It confirms that when it comes to preserving sight, the most effective treatment is the one the patient can afford to continue. The BRIO study provides the evidence necessary to make that treatment a reality for thousands, if not millions, of patients worldwide.
Study Citation:
Chakraborty, D.; Sinha, T.K.; Sinha, S.; Biswas, R.K.; Das, A.; Maiti, A.; Bhattacharya, R.; Dan, S.; Rungta, D.; Das, S. Real-World Comparison of Biosimilar Ranibizumab (Ranieyes) and Innovator Ranibizumab (Lucentis/Accentrix) Across Multiple Retinal Vascular Diseases (The BRIO Study). Pharmaceuticals 2026, 19, 747. https://doi.org/10.3390/ph19050747