In the evolving landscape of oncology, the integration of Artificial Intelligence (AI) into histopathology is marking a significant departure from traditional, subjective diagnostic methods. A pioneering study published in the journal Diagnostics (2026) has introduced a novel approach to risk stratification for Marginal Zone Lymphoma (MZL), potentially reshaping how clinicians manage this heterogeneous group of B-cell cancers. By utilizing AI-assisted quantitative immunohistochemical (IHC) analysis, researchers have successfully identified biomarkers that not only predict patient outcomes but also offer a path toward a more accurate prognostic model.
Main Facts: Redefining MZL Risk Assessment
Marginal Zone Lymphoma is the second most common subtype of indolent B-cell non-Hodgkin lymphoma, yet its clinical behavior remains notoriously difficult to predict. Current diagnostic tools, such as the MZL International Prognostic Index (MZL-IPI) and Ann Arbor staging, have historically struggled to pinpoint which patients are at high risk for "histologic transformation"—a process where an indolent lymphoma evolves into a much more aggressive form, such as diffuse large B-cell lymphoma (DLBCL).

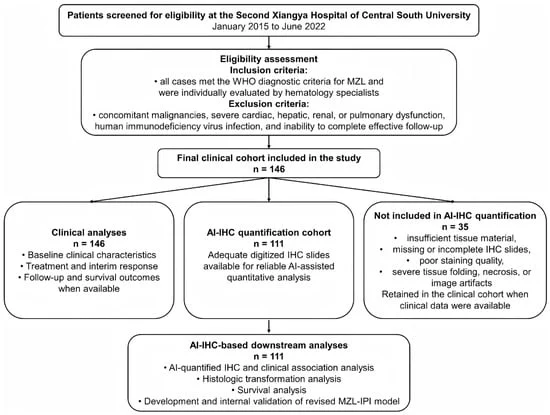
The study, led by researchers at the Second Xiangya Hospital of Central South University, analyzed 146 patients treated between 2015 and 2022. By applying deep learning algorithms to digitize and quantify 13 different IHC markers, the research team identified that specific protein expressions, notably CD3 and CD21, serve as powerful independent predictors of patient survival and disease progression. The primary breakthrough is the "CD21-revised MZL-IPI," a modified model that incorporates AI-derived data to provide a more granular view of patient risk than clinical metrics alone.
Chronology: From Data Collection to Model Validation
The research project followed a rigorous, multi-year developmental timeline designed to ensure both clinical relevance and statistical robustness:

- January 2015 – June 2022 (Data Collection): Clinical data and pathological specimens were gathered from 146 patients diagnosed with MZL.
- Methodological Standardization: Researchers employed an Intelligent Pathology Stereoscopic Analyzer to scan tissue samples. Of the original cohort, 111 patients provided high-quality digital slides suitable for AI-assisted quantitative analysis.
- March 25, 2026: The formal submission of the research findings.
- May 1, 2026: Peer-reviewed revisions were completed, addressing the integration of AI markers into existing prognostic indices.
- May 11, 2026: The study was officially published, offering a new framework for integrating AI-derived tissue biomarkers into clinical practice.
Supporting Data: The Power of CD3 and CD21
The strength of the study lies in its quantitative approach to markers previously evaluated only through qualitative observation. The AI system analyzed the percentage of positive cells within the tumor microenvironment, revealing distinct correlations:
- CD3 and Histologic Transformation: The study identified that low expression of CD3 (below 25.60%) is a significant independent predictor of histologic transformation. This suggests that reduced T-cell infiltration may signal a failure in the body’s immune surveillance, allowing the lymphoma to progress to a more malignant state.
- CD21 and Overall Survival (OS): High expression of CD21 was identified as a favorable prognostic factor. CD21 is essential for maintaining follicular dendritic cell (FDC) networks; its high presence suggests a more organized immune architecture within the tumor, which correlates with better long-term survival.
- Model Performance: Through 100,000 internal random resamplings and three-fold cross-validation, the revised MZL-IPI model consistently outperformed the original index. The C-index—a measure of predictive accuracy—showed that incorporating CD21 significantly sharpened the model’s ability to predict Overall Survival across various clinical subgroups, including patients undergoing different treatment modalities.
Official Perspectives: The Role of AI in Pathology
The authors, including senior researchers Yi Jiang and Hongling Peng, emphasize that this research is not intended to replace the pathologist’s expertise, but rather to augment it. "AI-assisted digital pathology is transforming histopathological assessment from traditional qualitative interpretation toward quantitative and reproducible analysis," the study notes.

The team highlights that while traditional clinical scoring systems like the MZL-IPI are useful, they often fail to capture the complexity of the tumor microenvironment. By digitizing the tissue, researchers can reduce inter-observer variability—a common issue in manual pathology—and provide continuous, objective data points that can be fed into predictive models. The study also acknowledges the collaborative efforts of technical partners, such as Changsha Lansi Intelligent Technology Co., Ltd., whose proprietary AI-pathology platform was utilized to standardize the digital image analysis.
Implications for Clinical Practice and Future Research
The implications of these findings are profound for the future of personalized oncology. If validated in larger, multi-center trials, the CD21-revised MZL-IPI could help clinicians make more informed decisions about treatment intensity. For example, patients identified as "high-risk" by the revised model might be candidates for more aggressive surveillance or earlier therapeutic intervention, while "low-risk" patients could potentially avoid the toxicity associated with intensive chemotherapy regimens.

However, the researchers maintain a cautious stance. Because the study was retrospective and based on a single institution’s data, they emphasize that the proposed thresholds—such as the 56.53% cut-off for CD21—are not yet "universal." They warn that these values are dependent on specific staining and scanning protocols and could vary if applied in different hospital settings.
"The revised MZL-IPI model proposed in this study should be considered an exploratory framework," the authors conclude. The path forward involves external, multi-center validation to ensure that the AI-derived markers perform consistently across different patient populations and technical environments. Furthermore, future studies are needed to explore the biological mechanisms behind why these specific T-cell and B-cell markers impact survival, potentially opening the door to new targeted immunotherapies.

In summary, this study represents a major step forward in bridging the gap between high-throughput digital imaging and clinical prognostic modeling. As AI continues to integrate into the diagnostic workflow, tools like the revised MZL-IPI may soon become standard, providing the precision necessary to navigate the complex, often unpredictable course of lymphoma.