Breakthrough in the Fight Against Drug-Resistant Malaria
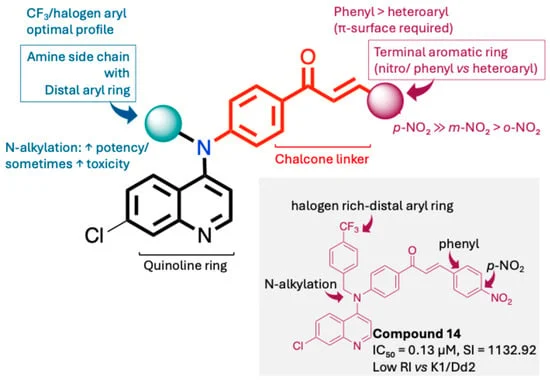
A team of researchers from the National Research Center for Protozoan Diseases at Obihiro University of Agriculture and Veterinary Medicine in Japan, in collaboration with North-West University in South Africa, has unveiled a new class of synthetic quinoline derivatives that show significant potential in combating chloroquine-resistant Plasmodium falciparum. The study, published in the journal Pharmaceuticals, highlights a specific lead compound—designated as Compound 14—that exhibits high antiplasmodial potency, exceptional selectivity, and, crucially, the ability to maintain effectiveness against strains of malaria that have evolved to evade traditional treatments.
As the global health community grapples with the persistent threat of malaria—a disease that continues to cause immense morbidity and mortality worldwide—the emergence of parasites resistant to conventional drugs like chloroquine has rendered many standard treatment regimens ineffective. This new research offers a glimmer of hope, providing a molecular blueprint for developing the next generation of antimalarial agents.
Chronology of Development
The development of these novel compounds was a multi-stage process spanning several years of rigorous chemical synthesis and biological evaluation.

- April 1, 2026: The research manuscript was formally submitted to the journal Pharmaceuticals, detailing the design and synthesis of eighteen quinoline derivatives incorporating arylnitro and aminochalcone moieties.
- April 29, 2026: Following a peer-review process, the study underwent revision to clarify the structure-activity relationship (SAR) analysis and the docking simulation protocols.
- May 4, 2026: The final version of the study was accepted for publication, having demonstrated consistent results in both sensitive and resistant P. falciparum strains.
- May 8, 2026: The study was published, providing the scientific community with a comprehensive analysis of the most potent derivative, Compound 14.
The research team, led by Yoshifumi Nishikawa, spent years exploring the hybridization of the quinoline core—the backbone of historic antimalarials—with chalcone systems. This "dual-function" approach was designed specifically to engage multiple targets within the parasite, aiming to bypass the resistance mechanisms that have historically plagued simpler quinoline-based drugs.
Supporting Data: A Closer Look at Compound 14
The study utilized a SYBR Green I-based growth inhibition assay to measure the effectiveness of the eighteen synthetic derivatives. The data revealed that several compounds exhibited sub-micromolar activity against the chloroquine-sensitive 3D7 strain. However, Compound 14 stood out as the most promising candidate.
Key Performance Metrics:
- Potency: Compound 14 achieved an IC$_50$ value of 0.13 µM against the 3D7 strain, far surpassing the moderate activity seen in other derivatives.
- Selectivity: The compound demonstrated a remarkable Selectivity Index (SI) of 1132.92, indicating it is highly toxic to the parasite while maintaining minimal toxicity toward human skin fibroblast (HSF) cells.
- Resistance Resilience: When tested against multidrug-resistant strains K1 and Dd2, Compound 14 showed resistance indices of 4.77 and 2.54, respectively. For comparison, chloroquine showed resistance indices of 28.46 and 34.42 against the same strains, confirming that the new compound is significantly more resilient to current parasite resistance mechanisms.
In addition to these laboratory results, molecular docking simulations provided a visual understanding of how Compound 14 interacts with its biological targets. The data suggests that the compound binds more extensively to the surface of hemozoin (the parasite’s toxic waste storage site) and to the P. falciparum chloroquine resistance transporter (PfCRT) than chloroquine does. By forming a broader network of hydrophobic and hydrogen-bonding interactions, the compound appears to "clog" the mechanisms that the parasite uses to survive exposure to traditional quinoline drugs.

Official Responses and Scientific Context
The research team emphasized that while the findings are groundbreaking, they represent a foundational step rather than an immediate cure. Dr. Yoshifumi Nishikawa, the corresponding author, noted that the study’s reliance on docking models—while highly predictive—serves as a framework for future drug development.
"The identification of Compound 14 provides a robust scaffold for further optimization," the study states. The authors acknowledge that while the compound is not yet perfectly "drug-like" according to strict pharmacological filters—due to its higher molecular weight—it successfully adheres to the Veber rule regarding molecular flexibility and polarity.
Independent experts have pointed out that the study’s use of both chloroquine-sensitive and multi-drug resistant strains (K1 and Dd2) adds significant weight to the findings. By demonstrating that the compound functions even when the PfCRT transporter is mutated, the researchers have addressed one of the most critical hurdles in contemporary malaria research.

Implications for Global Health
The implications of this study for the future of malaria treatment are profound. As the efficacy of current frontline treatments wanes, the need for compounds that can "reset" the standard of care is paramount.
Potential for Clinical Evolution
If Compound 14 or its derivatives can be successfully translated into clinical trials, they could provide a new tool for regions where resistance is rampant. The ability to retain activity against the Dd2 strain is particularly significant, as this strain is notoriously difficult to treat.
The Road Ahead
The researchers have identified several key areas for future work:

- In Vivo Efficacy: The immediate next step is to test the compound in animal models of malaria to determine its pharmacokinetic profile and actual therapeutic efficacy in a living system.
- Structural Optimization: Efforts will likely focus on reducing the molecular weight of the lead compound to improve its drug-like properties while maintaining the potent binding affinity observed in the laboratory.
- Mechanism Verification: While docking studies indicate a high probability of interference with hemozoin formation and PfCRT function, further biophysical studies are required to confirm these mechanisms experimentally.
Furthermore, the study addresses a secondary safety concern: hemolytic activity. All eighteen compounds were tested for their impact on human red blood cells, and the results were highly favorable, with negligible hemolysis rates. This suggests that the compound is unlikely to cause the erythrocyte membrane damage that can sometimes occur with experimental antimalarial drugs.
Conclusion
The work of Ariefta, Beteck, Legoabe, and Nishikawa represents a significant contribution to the field of medicinal chemistry. By integrating synthetic chemistry, in vitro parasite assays, and in silico docking, the team has successfully identified a promising lead that could eventually overcome the limitations of classic antimalarials.
While the researchers remain cautious—noting that "Compound 14" is not yet a clinical drug—their work provides a clear, data-driven pathway for the development of next-generation quinoline-based agents. As the world continues to push toward the goal of malaria eradication, such innovative approaches to overcoming drug resistance are not just beneficial; they are an absolute necessity. The scientific community will now be looking to see if these promising laboratory results can be successfully translated into the next generation of life-saving therapies.