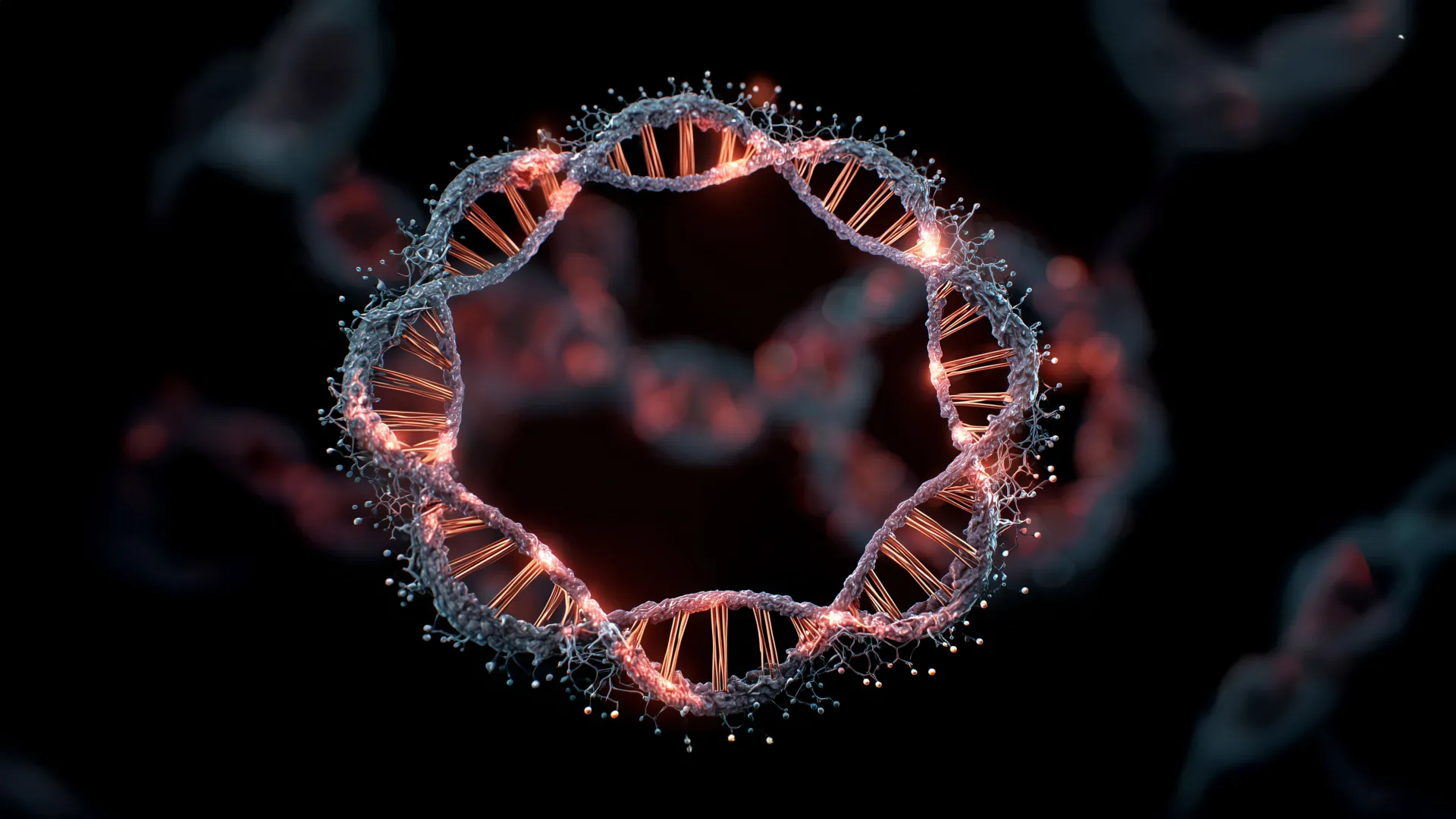
London, UK & Stanford, US – September 8, 2023 – In a landmark scientific breakthrough offering a vital new weapon against one of the most formidable cancers, an international consortium of scientists has unveiled a critical mechanism driving the aggressive growth of glioblastoma, the most common and deadliest adult brain cancer. Their discovery zeroes in on "extrachromosomal DNA" (ecDNA) – enigmatic rings of genetic material that operate independently of a cell’s main chromosomes – revealing them as potent, early-stage architects of glioblastoma’s devastating progression. This profound insight could revolutionize the way this formidable disease is diagnosed, tracked, and treated, opening previously unimaginable doors to earlier intervention and more effective therapies.
Published today in the prestigious journal Cancer Discovery, the findings are the first to definitively link the early emergence of ecDNA rings, laden with cancer-driving genes, to the initial stages of glioblastoma’s development. Crucially, in some cases, these rogue DNA structures were found to be present even before a discernible tumour had fully formed. This precocious appearance, the researchers suggest, doesn’t just precede the cancer; it actively orchestrates the conditions for its rapid proliferation, remarkable adaptability, and notorious resistance to conventional treatments.
The collaborative effort was spearheaded by Dr. Benjamin Werner, a distinguished group leader at Queen Mary University of London’s Barts Cancer Institute, and Professor Paul Mischel, the Fortinet Founders Professor at Stanford University. Both are integral members of team eDyNAmiC, a formidable international alliance funded by the Cancer Grand Challenges initiative. Contributing significantly to this pioneering work was also Professor Charlie Swanton, Deputy Clinical Director at The Francis Crick Institute. Their combined expertise, spanning oncology, evolutionary biology, computational science, and clinical research, has illuminated a previously dark corner of cancer biology.
The Main Facts: A Paradigm Shift in Understanding Glioblastoma
At its core, this research challenges long-held assumptions about how glioblastoma initiates and evolves. Instead of a gradual accumulation of genetic mutations within the main chromosomes, the study points to ecDNA as a primary, early-stage driver. These small, circular fragments of DNA, distinct from the linear chromosomes, are not merely bystanders in cancer development; they are active participants, capable of rapidly amplifying cancer-causing genes.
The key takeaway is the timing: ecDNA’s appearance is not a late-stage adaptation but an initiating event. This early arrival provides a fertile ground for the cancer’s aggressive nature, allowing it to bypass traditional cellular controls and rapidly evolve. The implications are profound, suggesting that targeting ecDNA could offer a novel strategy to disrupt glioblastoma at its very inception, fundamentally altering its trajectory.
Chronology: Tracing the Early Roots of an Aggressive Cancer
Glioblastoma stands as one of oncology’s most intractable challenges. With a median survival rate stubbornly hovering around 14 months, and little significant improvement in treatment outcomes over recent decades, the urgency for new approaches is palpable. The disease’s inherent heterogeneity, rapid growth, and ability to infiltrate surrounding brain tissue make it notoriously difficult to treat, often rendering even aggressive surgical and chemoradiation therapies ultimately ineffective. Patients face a grim prognosis, and the quality of life after diagnosis can deteriorate rapidly.
The scientific community has long grappled with understanding the fundamental mechanisms that endow glioblastoma with such a devastating capacity. While traditional genomic studies have focused on mutations within the main chromosomes, the role of ecDNA has, until recently, remained largely mysterious. These "rogue rings" of DNA were initially considered anomalies, but mounting evidence across various cancer types, including adult and paediatric cancers, has highlighted their potential as critical players.
Recognizing this burgeoning frontier, the Cancer Grand Challenges initiative – a bold, global funding partnership established by Cancer Research UK and the National Cancer Institute in the US – identified understanding ecDNA as one of the most formidable hurdles facing modern cancer research. In 2022, they committed $25 million to fund team eDyNAmiC. This ambitious, international, and cross-disciplinary consortium comprises experts from diverse fields – cancer biology, clinical research, evolutionary biology, computer science, and mathematics – united by the singular goal of deciphering ecDNA’s intricate role and identifying actionable strategies to target it. The current study represents a significant milestone in team eDyNAmiC’s mission, providing crucial insights into the evolutionary dynamics of glioblastoma.
To unravel the complex history of ecDNA in glioblastoma, team eDyNAmiC and their collaborators adopted an innovative approach, akin to forensic archaeology. Rather than relying on single biopsies, which offer only a snapshot in time, the researchers meticulously "excavated" multiple sites within and around glioblastoma tumours from patients. This multi-region sampling provided a rich, spatially resolved dataset of genomic and imaging information.
"We studied the tumours much like an archaeologist would," explains Dr. Benjamin Werner, senior author of the study and a group leader at the Barts Cancer Institute, Queen Mary University of London. "Rather than taking a single sample, we excavated multiple sites around the tumour, allowing us to build computational models describing how they evolved. We simulated millions of different scenarios to reconstruct how the earliest ecDNAs emerged, spread, and drove tumour aggressiveness, giving us a clearer picture of the tumour’s origins and progression."
This sophisticated computational modeling was paramount. By simulating countless evolutionary pathways, the researchers could rewind the clock, piecing together the chronological emergence and expansion of ecDNA. This deep dive into the tumour’s past revealed a startling truth: ecDNA isn’t just present; it’s a foundational element in the cancer’s evolutionary architecture, arriving remarkably early in the disease’s timeline.
Supporting Data: Unmasking the EGFR Driver and a Window of Opportunity
The comprehensive analysis yielded striking results, consistently pointing to one particular gene as a dominant force within these rogue ecDNA rings: EGFR (Epidermal Growth Factor Receptor). EGFR is a well-known oncogene, a gene with the potential to cause cancer, and its overexpression or mutation is frequently implicated in various malignancies. However, its presence on ecDNA, and particularly its early appearance, introduces a new dimension to its role in glioblastoma.
The study revealed that most ecDNA rings detected contained EGFR, highlighting its central role in driving the disease. Crucially, the researchers found that EGFR ecDNA appeared remarkably early in the cancer’s evolution – in some patients, even preceding the formation of a fully established tumour. This early presence acts as a powerful accelerant, conferring a significant growth advantage to nascent cancer cells.
Furthermore, the research uncovered that these EGFR-laden ecDNAs frequently acquired additional genetic alterations, such as the EGFRvIII variant. This specific variant is notorious for making cancers even more aggressive and, critically, more resistant to a wide array of existing therapies. The sequential acquisition of these changes on ecDNA paints a vivid picture of a highly adaptable and rapidly evolving cancer, driven by these extrachromosomal elements.
This detailed understanding of ecDNA’s evolutionary trajectory opens up a tantalizing prospect: a "window of opportunity" for earlier intervention. "These subtle mechanisms show that there may be a window of opportunity to detect and treat the disease between the first appearance of EGFR ecDNA and the emergence of these more aggressive variants," suggests Dr. Magnus Haughey, a postdoctoral researcher in Dr. Werner’s group and one of the paper’s lead authors.
The implication is profound: if scientists can develop a reliable, non-invasive test capable of detecting early EGFR ecDNA – for instance, through a simple blood test (often referred to as a "liquid biopsy") – it could enable clinicians to intervene at a stage when the disease is potentially less aggressive and more amenable to treatment. This early detection could transform the current bleak prognosis for glioblastoma patients, shifting the focus from managing advanced disease to intercepting it before it becomes intractable.
Beyond EGFR, the study also confirmed that ecDNA can carry more than one cancer-driving gene simultaneously. This multi-gene cargo capability further complicates the tumour’s biology, as each unique combination of genes on ecDNA can uniquely shape how the tumour evolves, responds to different treatments, and develops resistance. This finding underscores the potential value of tailoring treatments based on a tumour’s specific ecDNA profile – a move towards truly personalized medicine for glioblastoma.
Despite these significant advancements, many mysteries surrounding ecDNA persist. The researchers are now charting their future course, planning to investigate how different therapeutic interventions impact the number and types of ecDNA present in glioblastoma cells. Team eDyNAmiC, in its broader mission, will continue to explore the role of ecDNAs across a spectrum of cancer types, aiming to unearth further opportunities for earlier diagnosis, more precise disease tracking, and the design of smarter, more targeted treatments across oncology.
Official Responses: Voices of Hope and Determination
The release of these findings has generated considerable excitement within the scientific and medical communities, with leading figures emphasizing the transformative potential of the research.
Dr. Benjamin Werner, senior author and group leader at Queen Mary University of London, reiterated the archaeological analogy, highlighting the depth of their investigation: "We studied the tumours much like an archaeologist would. Rather than taking a single sample, we excavated multiple sites around the tumour, allowing us to build computational models describing how they evolved. We simulated millions of different scenarios to reconstruct how the earliest ecDNAs emerged, spread, and drove tumour aggressiveness, giving us a clearer picture of the tumour’s origins and progression." His words underscore the meticulous and innovative methodology employed, which was critical to unearthing these hidden evolutionary dynamics.
Dr. Magnus Haughey, a lead author and postdoctoral researcher in Dr. Werner’s group, articulated the immediate clinical potential: "These subtle mechanisms show that there may be a window of opportunity to detect and treat the disease between the first appearance of EGFR ecDNA and the emergence of these more aggressive variants. If scientists can develop a reliable test to detect early EGFR ecDNA – for example through a blood test – it could enable them to intervene before the disease becomes harder to treat." His statement directly links the scientific discovery to tangible patient benefits.
Professor Charlie Swanton, Deputy Clinical Director and head of the Cancer Evolution and Genome Instability Laboratory at The Francis Crick Institute and chief clinician at Cancer Research UK, expressed profound optimism regarding the implications for patient care: "These findings suggest that ecDNA is not just a passenger in glioblastoma, but an early and powerful driver of the disease. By tracing when and how ecDNA arises, we open up the possibility of detecting glioblastoma much earlier and intervening before it becomes so aggressive and resistant to therapy. I hope this might help to drive a new era in how we diagnose, track and treat this devastating cancer." Professor Swanton’s vision of a "new era" encapsulates the scale of this breakthrough.
Paul Mischel, MD, the Fortinet Founders Professor and professor and vice chair of research in the pathology department at Stanford Medicine, provided further context on ecDNA’s broader role in cancer and the specific significance for glioblastoma: "These findings reveal an important new insight into the role of ecDNA in tumour development and progression. Previous work from our collaborative team and other researchers, has shown that ecDNA can arise early in tumor development, including at the stage of high-grade dysplasia, and it can also arise later to drive tumor progression and treatment resistance. The findings here show that in glioblastoma, there is an early event driven by ecDNA that could potentially be more actionable, raising the possibility that glioblastoma is another cancer for which earlier detection and intervention based upon ecDNA may be possible." Dr. Mischel’s comments highlight the evolving understanding of ecDNA and position this glioblastoma research within a larger scientific narrative.
Finally, Dr. David Scott, Director of Cancer Grand Challenges, lauded the collaborative spirit and bold vision of the initiative: "This study exemplifies the bold, boundary-pushing science Cancer Grand Challenges was created to support. By unravelling the evolutionary history of ecDNA in glioblastoma, team eDyNAmiC is not only deepening our understanding of one of the most devastating cancers but also illuminating new paths for earlier detection and treatment. It’s a powerful reminder that when we bring together diverse disciplines and global talent, we can begin to solve the toughest problems facing cancer research." Dr. Scott’s statement emphasizes the power of interdisciplinary collaboration in tackling grand challenges.
Implications: Paving the Way for a New Era in Brain Cancer Treatment
The implications of this groundbreaking research extend far beyond the laboratory, offering a beacon of hope for patients and clinicians grappling with glioblastoma. The identification of ecDNA as an early and potent driver fundamentally shifts the paradigm of glioblastoma research and treatment.
Early Detection and Prevention: The most immediate and transformative implication is the potential for earlier diagnosis. If a reliable blood test can be developed to detect EGFR ecDNA, it could allow for intervention long before a tumour becomes clinically apparent or aggressively advanced. This could mean detecting the disease when it is still in its nascent, potentially less aggressive stages, opening up possibilities for preventative strategies or treatments with significantly higher efficacy. Imagine a world where glioblastoma could be detected through a routine screening, much like other cancers, before it has a chance to take root and spread.
Precision Medicine: The finding that ecDNA can carry multiple cancer genes, and that its profile influences tumour evolution and treatment response, paves the way for highly personalized treatment strategies. By profiling a patient’s tumour for its specific ecDNA cargo, clinicians could tailor therapies, selecting drugs most likely to be effective against the unique genetic drivers present. This move away from a one-size-fits-all approach promises to enhance treatment effectiveness and minimize adverse side effects.
Targeted Therapies: Understanding how ecDNA drives glioblastoma growth provides new targets for drug development. Pharmaceutical companies and academic researchers can now focus on designing therapies that specifically inhibit the formation, replication, or function of these rogue DNA rings, or target the specific genes like EGFR that they carry, particularly when they appear early.
Tracking Disease Progression and Resistance: Beyond initial diagnosis, monitoring ecDNA levels and profiles could offer a powerful tool for tracking disease progression and predicting treatment resistance. A rise in aggressive ecDNA variants could signal a need to switch therapies, allowing for proactive adjustments to treatment plans rather than reactive responses to clinical deterioration.
Broader Cancer Impact: While this study focuses on glioblastoma, the principles unearthed here have broader implications for other cancers where ecDNA is known to play a role. The methodology, the understanding of ecDNA’s early emergence, and the potential for ecDNA-based diagnostics and therapeutics could be transferable to a wide array of adult and paediatric malignancies. Team eDyNAmiC’s ongoing efforts to investigate ecDNAs across various cancer types are testament to this expansive vision.
In conclusion, this discovery marks a pivotal moment in the fight against glioblastoma. By peeling back the layers of this devastating disease, scientists have not only revealed a hidden driver but also illuminated a clear path forward. The journey from this groundbreaking research to widespread clinical application will undoubtedly be challenging, requiring sustained investment, continued collaboration, and rigorous testing. However, the promise of earlier detection, smarter treatments, and ultimately, a more hopeful future for glioblastoma patients, makes this pursuit an imperative for global health. This research is not merely a scientific publication; it is a profound declaration of intent – a commitment to transforming the landscape of brain cancer care.