Ann Arbor, MI – A groundbreaking study published in the journal Cancers reveals a critical interplay between tumor hypoxia and the tumor microenvironment (TME) in orchestrating the demise of crucial anti-cancer immune cells, specifically CD8+ T cells, within pancreatic ductal adenocarcinoma (PDAC). This research sheds light on why pancreatic cancer has remained a formidable challenge for immunotherapies and suggests potential avenues for improving treatment strategies.
The study, led by Ashley M. Mello, Marina Pasca di Magliano, and Kyoung Eun Lee from the University of Michigan, elucidates how the oxygen-deprived conditions prevalent in PDAC tumors, combined with signals from cancer cells and supportive fibroblasts (cancer-associated fibroblasts or CAFs), conspire to cripple CD8+ T cells. This debilitating effect manifests as suppressed proliferation, increased cell death, and the adoption of a dysfunctional, terminally exhausted state, ultimately hindering the immune system’s ability to combat the cancer.

Unraveling the Complex Microenvironment of Pancreatic Cancer
Pancreatic ductal adenocarcinoma (PDAC) is notorious for its aggressive nature and poor prognosis, often characterized by its dense, stroma-rich architecture and severely hypoxic interior. These features contribute significantly to its resistance against various cancer therapies, including the promising field of immunotherapy. Immunotherapies aim to harness the patient’s own immune system to fight cancer, with CD8+ T cells being central players in recognizing and eliminating tumor cells. However, in the hostile environment of PDAC, these vital immune warriors often fail to mount an effective response.
The research team sought to dissect the intricate mechanisms by which the PDAC TME undermines CD8+ T cell function. They hypothesized that both the intrinsic effects of hypoxia on T cells and the extrinsic signals emanating from the surrounding tumor and stromal cells play crucial roles.

Key Findings: A Multi-Pronged Attack on CD8+ T Cells
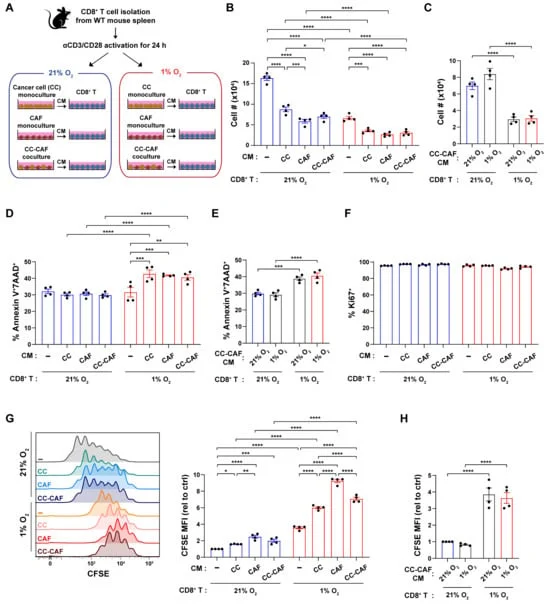
The study employed a sophisticated in vitro model system that mimicked the conditions found within a PDAC tumor. This involved culturing CD8+ T cells in the presence of conditioned media (CM) derived from pancreatic cancer cells and CAFs, under both normoxic (normal oxygen) and hypoxic (low oxygen) conditions.
1. Suppression of Proliferation and Survival: The research demonstrated that hypoxia alone significantly reduced the proliferation of CD8+ T cells. When combined with factors secreted by pancreatic cancer cells and CAFs, this suppressive effect was amplified, leading to a marked increase in CD8+ T cell death. While neither hypoxia nor the secreted factors alone were sufficient to cause substantial cell death, their synergistic action proved detrimental to T cell survival.

2. Modulation of Effector Functions: While hypoxia, in conjunction with tumor and CAF signals, did enhance the expression of certain effector molecules like granzyme B (GZMB) on a per-cell basis, this was overshadowed by the reduced number of viable T cells. The overall pool of functional effector T cells was significantly diminished. Furthermore, the study observed a complex interplay in the regulation of Interferon-gamma (IFN-γ). Hypoxia alone suppressed IFN-γ production, while CM from tumor and CAF co-cultures could further inhibit it. Interestingly, the specific factors secreted by tumor and CAF cells under hypoxic conditions appeared to have a more potent suppressive effect on IFN-γ compared to those produced under normoxia.
3. Upregulation of Co-Inhibitory Receptors: The study also revealed that hypoxia and the signals from tumor and CAF cells cooperatively led to the upregulation of co-inhibitory receptors on CD8+ T cells. These receptors, such as PD-1, TIM-3, and LAG-3, act as "brakes" on T cell activity, preventing them from attacking cancer cells. The combined effect of hypoxia and TME-derived factors essentially silenced the anti-tumor response by engaging these inhibitory pathways.

4. Evidence from Human Pancreatic Tumors: To validate their in vitro findings, the researchers analyzed single-cell RNA sequencing (scRNA-seq) data from human PDAC tumors. This analysis confirmed that CD8+ T cells found in hypoxic regions of human tumors exhibited signatures associated with increased apoptosis (programmed cell death) and stress responses. Crucially, these hypoxic T cells also displayed reduced expression of genes associated with stem-like properties and progenitor T cell function, while simultaneously showing increased expression of genes linked to terminal differentiation and exhaustion. This suggests that hypoxia drives CD8+ T cells towards a state of irreversible dysfunction.
Implications for Immunotherapy
The findings of this study have profound implications for the development and application of immunotherapies for pancreatic cancer. The dense, hypoxic TME of PDAC creates a hostile environment that actively suppresses the anti-tumor immune response. This explains, in part, why many patients with pancreatic cancer do not respond to current immunotherapeutic agents.

The research highlights that strategies aimed at improving immunotherapy efficacy in PDAC may need to address the dual challenges of hypoxia and the immunosuppressive signals from the tumor and stromal components. Potential therapeutic approaches could include:
- Hypoxia-Inducing Agents: Developing treatments that can reduce tumor hypoxia, thereby creating a more permissive environment for T cell function.
- Targeting TME-Derived Factors: Identifying and neutralizing the specific immunosuppressive molecules secreted by cancer cells and CAFs that impair T cell activity.
- Combination Therapies: Combining immunotherapies with agents that target hypoxia or the immunosuppressive stroma to synergistically enhance anti-tumor immunity.
- Reprogramming T Cell States: Investigating ways to prevent CD8+ T cells from entering the terminally exhausted state or to reverse this exhaustion.
Future Directions and Conclusion
The study’s authors emphasize the need for further research to pinpoint the precise molecular mechanisms by which hypoxia and TME factors interact to promote T cell dysfunction. Identifying the specific proteins and metabolites involved could pave the way for novel therapeutic targets. Moreover, in vivo studies are crucial to confirm these findings in a more complex biological setting and to understand how the spatial heterogeneity of the TME influences T cell responses.

In conclusion, this comprehensive study provides compelling evidence that hypoxia is not merely a consequence of PDAC but an active driver of CD8+ T cell dysfunction. By crippling these critical immune cells and pushing them towards a terminally exhausted state, the hypoxic TME significantly undermines the potential of immunotherapies. Understanding and overcoming these challenges is paramount to unlocking new and more effective treatment strategies for pancreatic cancer patients.
Article Details:
- Hypoxia Impairs CD8+ T Cell Fitness and Is Associated with a Dysfunctional CD8+ T Cell State in Pancreatic Cancer
- Authors: Ashley M. Mello, Marina Pasca di Magliano, Kyoung Eun Lee
- Journal: Cancers
- Volume: 18
- Issue: 10
- Article Number: 1508
- DOI: 10.3390/cancers18101508
- Publication Date: May 8, 2026