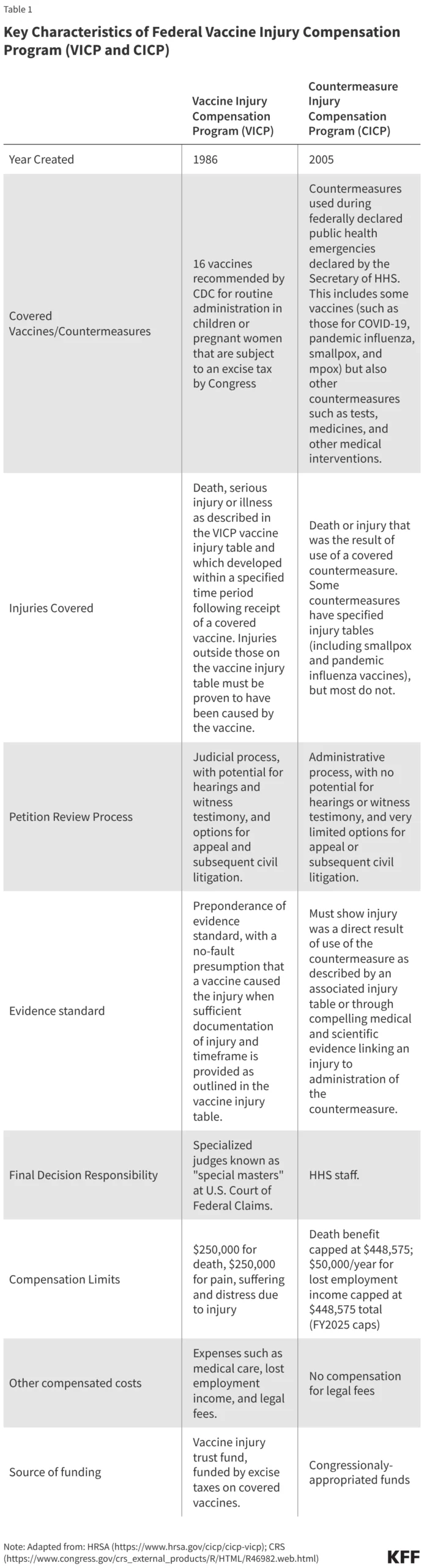
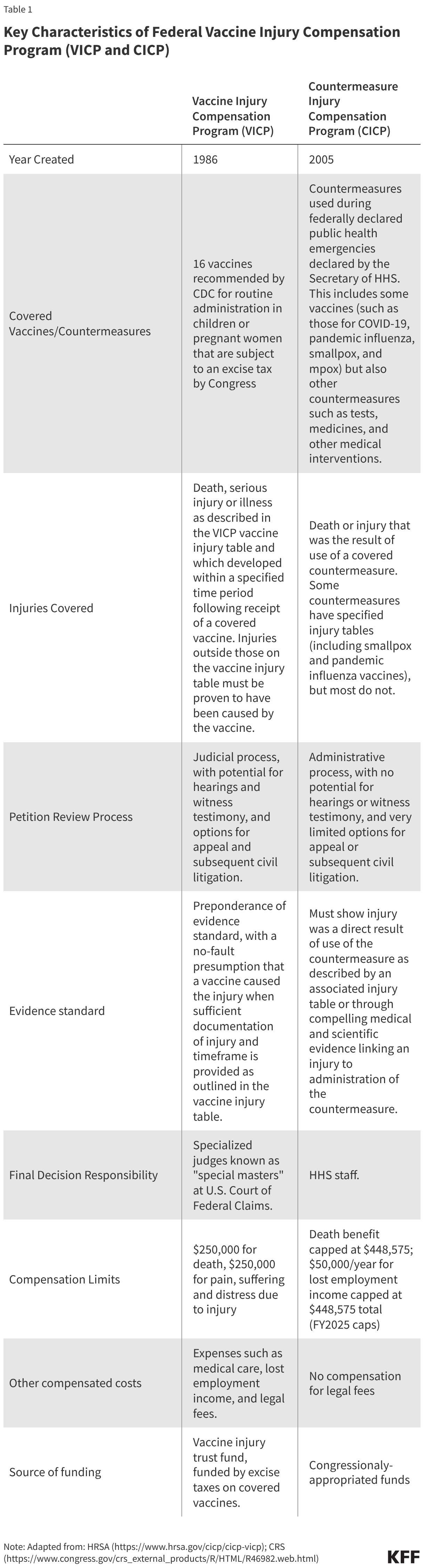
For decades, the United States has operated two distinct federal programs designed to act as a safety net for individuals who suffer rare adverse reactions to vaccinations. The National Vaccine Injury Compensation Program (VICP) and the Countermeasures Injury Compensation Program (CICP) serve a dual purpose: they provide a non-adversarial path for claimants to seek redress for injuries, and they insulate the vaccine market from the volatility of civil litigation that threatened to destabilize public health supply chains in the late 20th century.
However, these programs—long considered administrative backwaters—have recently been thrust into the center of a heated political debate. As calls for reform intensify, the future of how the U.S. government handles vaccine-related harm remains uncertain, sparking fears among public health experts that structural changes could undermine vaccine confidence and the stability of the pharmaceutical market.
The Origins and Rationales: Why the Programs Exist
The existence of these programs is rooted in the "Vaccine Crisis" of the early 1980s. During that period, the U.S. vaccine industry faced a wave of litigation that led manufacturers to signal their exit from the market. Faced with the prospect of losing the ability to provide essential childhood immunizations, Congress intervened.
The VICP was established in 1986 under the National Childhood Vaccine Injury Act. It was designed as a "no-fault" system. Instead of suing manufacturers in civil court—where legal fees are high and outcomes unpredictable—injured parties can file a petition with the U.S. Court of Federal Claims. The goal was to provide rapid, fair compensation while ensuring that vaccine makers remained willing to produce the products necessary for public health.
The CICP, by contrast, was established later under the Public Readiness and Emergency Preparedness (PREP) Act. Its scope is narrower, designed specifically for injuries resulting from "countermeasures" (like vaccines or medications) administered during a declared public health emergency. Unlike the VICP, which is a quasi-judicial system, the CICP is an administrative process run by the Department of Health and Human Services (HHS).

Chronology of Crisis and Criticism
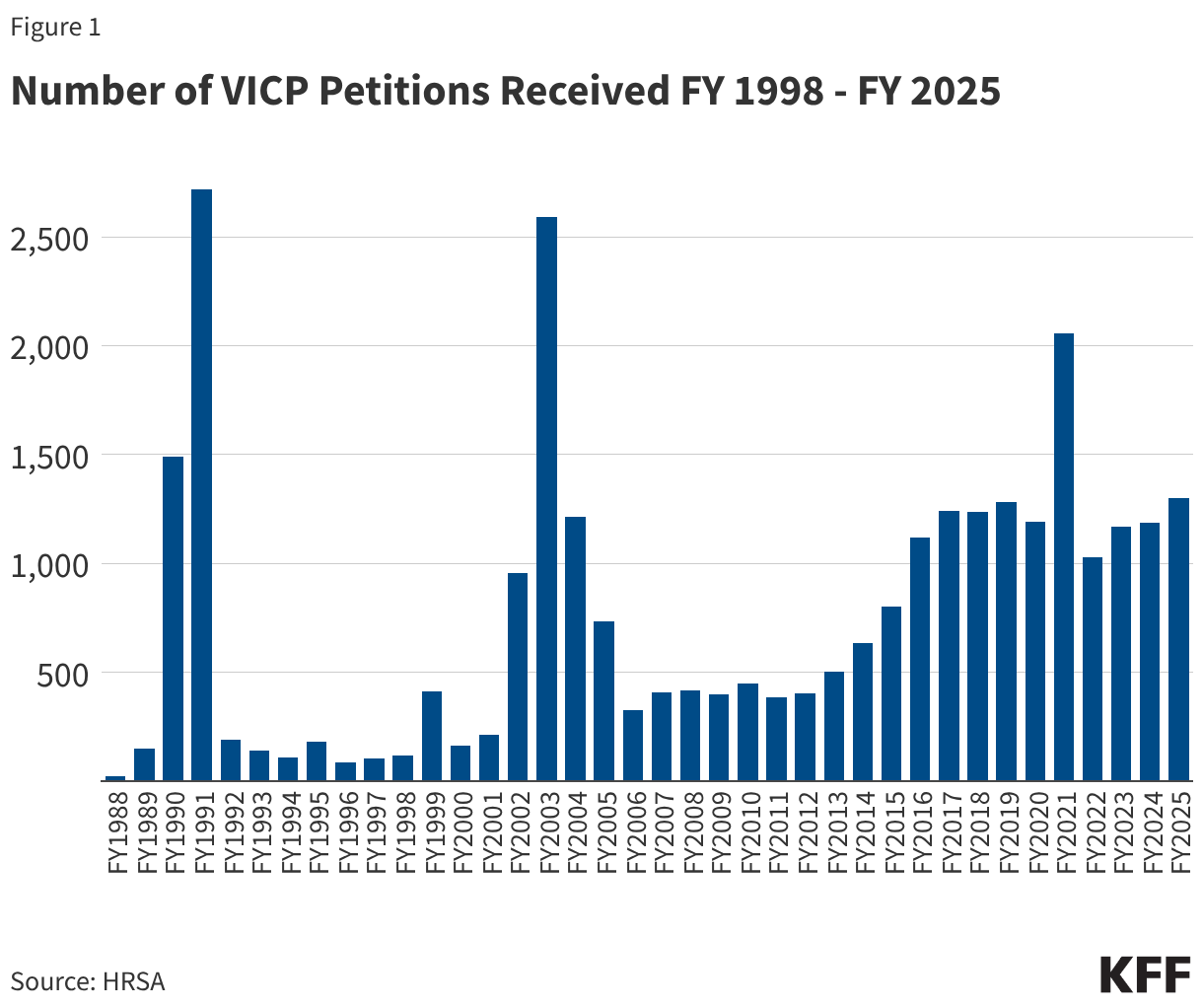
The political landscape surrounding these programs shifted dramatically in 2025. Secretary of Health and Human Services Robert F. Kennedy Jr. labeled the VICP a "morass of inefficiency, favoritism, and outright corruption." These accusations, though lacking in specific evidentiary support, signaled a pivot toward a more aggressive, reform-minded administration.
- 1986: VICP is established, creating a specialized "vaccine court" to manage liability.
- 2005: The PREP Act authorizes the CICP, creating a mechanism for pandemic-era liability.
- 2021: The COVID-19 pandemic leads to a massive surge in claims filed under the CICP, as millions receive novel mRNA and viral vector vaccines.
- 2025: High-level administration officials call for a complete overhaul of the existing compensation frameworks, citing systemic failures.
- 2026: Bipartisan legislation is introduced by members of Congress seeking to either eliminate or radically restructure these programs, reflecting growing legislative impatience with the status quo.
Supporting Data: By the Numbers
The disparity between the two programs is best highlighted by their respective success rates and financial structures. While both programs aim to compensate the injured, they operate under vastly different mandates.
The VICP Performance
The VICP is a more mature, well-funded system. Historically, it has compensated a significant portion of its petitions. Because it is funded by an excise tax on vaccines, it possesses a dedicated revenue stream that allows for larger, more comprehensive awards, including coverage for pain and suffering and medical expenses. Data indicates that VICP compensates roughly 48% of its petitions, a high success rate compared to typical civil litigation.
The CICP Performance
The CICP faces significantly more scrutiny. Since the introduction of COVID-19 vaccines, the volume of petitions has skyrocketed. However, the program has a remarkably low success rate; less than 1% of COVID-19 vaccine-related claims are found to be compensable. This is largely because the CICP requires a much higher evidentiary burden and does not offer the same legal protections or compensation categories as the VICP.
Comparative Efficiency
Relative to the hundreds of millions of vaccine doses administered in the U.S., the number of compensable petitions is statistically negligible.
- VICP: Approximately 1.89 compensable petitions per million doses.
- CICP: Approximately 0.14 compensable claims per million COVID-19 doses.
Processes for Review: How Claims are Adjudicated
The mechanics of these programs are fundamentally different.
VICP Process:
- Petition: A claimant files a petition with the U.S. Court of Federal Claims.
- HHS Review: The Department of Health and Human Services reviews the medical evidence.
- Special Master: A "Special Master" (a judge-like official) evaluates the claim.
- Adjudication: The court determines if the vaccine caused the injury, often relying on a "Vaccine Injury Table" that lists specific conditions associated with certain vaccines.
CICP Process:
- Administrative Filing: The claimant submits a request to the Health Resources and Services Administration (HRSA).
- Internal Adjudication: The claim is reviewed by internal staff within HHS.
- Limited Recourse: There is no "vaccine court" equivalent; the process is entirely administrative.
- Caps: The program is limited by annual appropriations, which critics argue keeps compensation amounts artificially low.
Most CICP awards remain under $10,000, whereas VICP awards frequently reach six-to-seven figures, reflecting the inclusion of long-term care costs.
Official Responses and Political Implications
The political discourse surrounding these programs is increasingly polarized. Proponents of the status quo argue that the current systems are a vital bulwark against the collapse of the vaccine market. They warn that if the VICP or CICP were abolished, manufacturers might cease production of essential vaccines to avoid the existential risk of class-action lawsuits.
Conversely, critics—including Secretary Kennedy—argue that the current systems are designed to protect manufacturers at the expense of patients. They advocate for a more transparent, claimant-friendly process that lowers the burden of proof.
The Legislative Tug-of-War
Several bills currently circulating in Congress suggest a divergence in strategy:
- The "Reformers": Seek to expand the Vaccine Injury Table, shorten wait times for adjudication, and increase compensation caps.
- The "Abolitionists": Argue that the government should not be involved in shielding manufacturers from liability, suggesting that a return to civil court would be a more "just" outcome for victims.
Implications: The Road Ahead
The implications of changing these programs are profound. If the U.S. shifts away from the VICP model, it risks creating a "litigation environment" that could cause pharmaceutical companies to exit the U.S. vaccine market. This would have an immediate impact on public health, potentially leading to shortages of childhood vaccines and a decline in immunization rates.
Furthermore, any shift in the compensation structure affects public trust. When the government appears to be denying claims or when it appears to be favoring industry over the injured, the "vaccine hesitancy" movement gains ammunition.
Key Policy Issues for Future Consideration:
- Transparency: How can HHS improve the visibility of the CICP decision-making process?
- Funding Stability: Should the CICP be moved toward a trust-fund model similar to the VICP to ensure consistent compensation?
- The Evidentiary Gap: Is it possible to bridge the gap between the strict scientific requirements for "causation" and the lived experience of vaccine-injured patients?
As the debate continues, the fundamental tension remains: how to balance the rights of the individual injured by a medical intervention with the collective need for a stable, accessible, and safe vaccine supply. The "morass" described by the current administration is, in the eyes of many, the inevitable friction of managing a public health necessity in a litigious society. Whether that system is "fixed" or "replaced" will likely define the landscape of American public health for the next generation.
Ultimately, the challenge for lawmakers is to implement reforms that enhance fairness and efficiency without inadvertently triggering a market exit by vaccine manufacturers—a scenario that would leave the American public far more vulnerable to infectious disease.