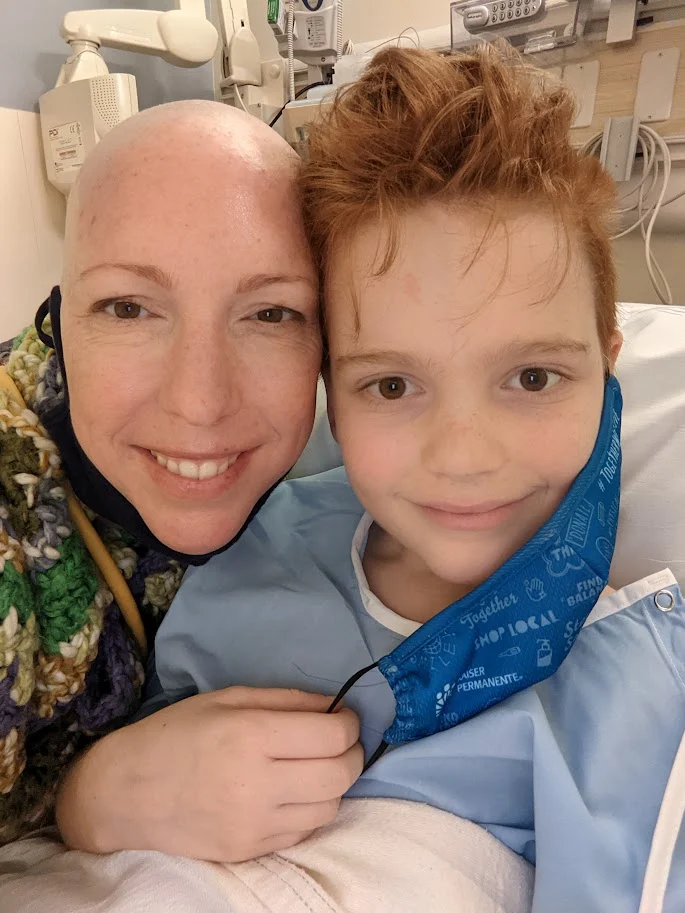
In the spring of 2012, Melinda Bachini checked into the National Cancer Institute (NCI) not as a mother, a wife, or a professional, but as “Patient 3737.” While the numerical designation might have felt impersonal to an outsider, for Bachini, it represented the most significant lifeline of her life. It was a formal entry into a high-stakes clinical trial—a final, desperate reach for survival after standard-of-care treatments had failed to halt the aggressive progression of her cholangiocarcinoma, or bile duct cancer.
Today, over a decade later, Bachini stands as a testament to the transformative power of medical research. Her journey from a stage IV diagnosis to a long-term survivor and current Chief Patient Officer at the Cholangiocarcinoma Foundation is more than a personal victory; it is a clarion call for a fundamental shift in how we perceive, fund, and participate in clinical trials.
The Chronology of a Survival Story
The trajectory of Bachini’s illness began with a brutal reality check. Cholangiocarcinoma is notoriously difficult to detect, often remaining silent until it reaches advanced stages. By the time of her diagnosis, the tumor in her liver had engulfed the entire right lobe.
Initially, surgery provided a temporary reprieve. However, the reprieve was short-lived. The cancer metastasized to her lungs, and conventional chemotherapy, the standard fallback for many patients, proved ineffective. By 2012, the situation had become dire. Bachini recalls the physical toll: her tumors were expanding with aggressive speed, leaving her breathless after climbing even a short flight of stairs.
It was during a sleepless night, amidst the mounting pressure of medical debt and the encroaching reality of a terminal prognosis, that Bachini took control of her own medical narrative. An exhaustive online search led her to the work of Dr. Steven Rosenberg and his pioneering team at the NCI. They were exploring “tumor-infiltrating lymphocyte (TIL) therapy,” a form of immunotherapy that essentially re-engineers the body’s own defenses.
“I applied—what did I have to lose?” Bachini reflects.
The treatment protocol was grueling. It involved sequencing her tumor to identify specific immune cells capable of recognizing and attacking the cancer. Once identified, these cells were extracted, harvested in a lab to multiply into the billions, and then re-infused into her body. It was a pioneering procedure, not yet proven for bile duct cancer, but it was a calculated risk that paid off. Within six months, her tumors showed significant regression; in the subsequent six months, they achieved a state of stability. That trial did more than buy her time—it gave her back her life.
The Stagnation of Trial Participation
Despite the success stories like Bachini’s, the clinical trial landscape remains plagued by low participation rates. Data from oncology research organizations indicate that only 5% to 7% of adult cancer patients enroll in clinical trials. This figure has remained stubbornly stagnant for years, even as the complexity and potential efficacy of these trials have increased exponentially.

The barriers to entry are multifaceted and systemic. For the average patient, the hurdle is often a lack of accessible information. Oncology care is frequently fragmented, and unless a patient is treated at a major academic research center, they may never be informed about relevant trial opportunities.
Beyond information gaps, geographic and financial barriers loom large. Many trials are conducted at centralized hubs, requiring patients to travel long distances, stay in hotels, and navigate complex insurance landscapes. Fear, too, plays a significant role. The pervasive, albeit outdated, myth that clinical trials are a “last resort” or that participants are merely “guinea pigs” continues to discourage patients from exploring options that could be their most effective path to remission.
The Urgent Need for Equitable Access
The issue of access is compounded by systemic inequities. Patients from marginalized and underrepresented communities face a “triple threat” of obstacles: geographic distance from major medical centers, cultural barriers in communication, and socioeconomic constraints that make the time-intensive nature of trial participation impossible.
When these populations are excluded, the medical community loses more than just potential participants; it loses vital data. Clinical trials are designed to answer complex questions about how different biological profiles respond to treatment. If a study cohort lacks diversity, the resulting findings may not be broadly applicable, potentially exacerbating health disparities.
“We are missing critical data,” Bachini asserts. “When we don’t have diverse representation in clinical trials, we aren’t just failing those individuals—we are failing to develop more effective treatments for everyone.”
Official Perspectives and the Industry Shift
Leading oncologists and health policymakers are increasingly echoing Bachini’s concerns. The medical establishment is slowly moving toward a decentralized model of clinical research. The goal is to move the trial to the patient, rather than requiring the patient to move to the trial.
Dr. Steven Rosenberg’s work, which saved Bachini’s life, is part of a broader shift toward personalized medicine. Federal agencies, including the FDA and the NCI, have begun incentivizing pharmaceutical companies to prioritize diversity in their trial design. New mandates require developers to submit a "Diversity Action Plan" for clinical studies, ensuring that demographic representation is considered from the early stages of protocol development.
However, advocates argue that these policy changes, while necessary, are not moving fast enough. There is a pressing need for:
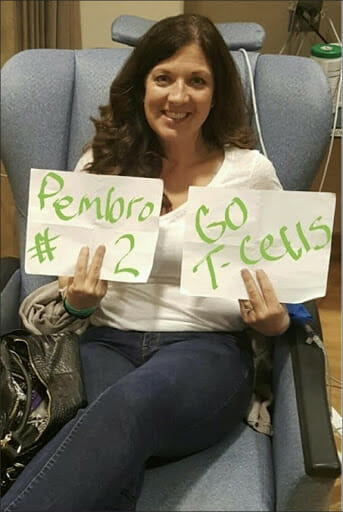
- Enhanced Provider Education: Oncologists in community settings must be equipped with the tools to screen patients for trials early in their treatment journey.
- Financial Support Models: Grants and assistance programs must be expanded to cover the travel, lodging, and childcare costs associated with trial participation.
- Simplified Enrollment: The bureaucracy of informed consent and screening must be streamlined without compromising safety.
The Implications of a New Paradigm
The implications of expanding clinical trial participation are profound. A successful trial does more than treat the individual; it creates a blueprint for future medicine. Every patient who participates in a trial contributes to the “collective knowledge” of the medical community. As Bachini notes, her own participation helped pave the way for a deeper understanding of how immunotherapy might be utilized in rare cancers like cholangiocarcinoma.
Furthermore, moving beyond the "last resort" stigma could shift the culture of cancer care. If patients and providers view clinical trials as a standard, high-quality treatment option rather than a desperate final measure, patients could access cutting-edge therapies much earlier in their treatment cycle, when their bodies are better equipped to handle the demands of the trial.
A Call to Action
For those currently navigating a cancer diagnosis, or for the families supporting them, the message is clear: knowledge is the most powerful tool. Patients should not hesitate to ask their oncologists about clinical trial options at every stage of their treatment, not just when standard options are exhausted.
“No one should ever be just a number,” Bachini says. “But every number has the potential to become a story of survival.”
As we look toward the future of oncology, the focus must remain on dismantling the silos that keep research and patients apart. Whether through increased funding for rare disease research, the adoption of telemedicine to bridge geographic gaps, or simply the courage to challenge the status quo, the goal is unified: to ensure that the next medical breakthrough is accessible to anyone, anywhere, who needs it.
The journey from Patient 3737 to advocate is not an anomaly; it is a blueprint. By transforming how we approach clinical trials, we can ensure that the next decade of cancer research is defined not just by the brilliance of the science, but by the inclusivity of the access.
Melinda Bachini serves as the Chief Patient Officer at the Cholangiocarcinoma Foundation. Her story is a reminder that while the road to a cure is complex, it is one that patients and scientists must walk together.