Nanjing, China – May 13, 2026 – A groundbreaking study published today in the journal Cancers has unveiled the potential of dedicated breast positron emission tomography (D-PET) combined with advanced artificial intelligence (AI) techniques to predict the success of neoadjuvant chemotherapy (NAC) in patients with HER2-positive breast cancer. This innovative approach could pave the way for more personalized and effective treatment strategies, minimizing unnecessary interventions and optimizing patient outcomes.
The Challenge of Predicting Treatment Response
HER2-positive breast cancer, a particularly aggressive subtype, often necessitates neoadjuvant chemotherapy administered before surgery. The goal is to shrink tumors, making surgical removal more feasible and increasing the likelihood of breast-conserving procedures. While achieving a pathological complete response (pCR)—the complete eradication of cancer cells in both the breast and lymph nodes—is a strong indicator of favorable long-term prognosis, reliably predicting which patients will achieve this outcome noninvasively has remained a significant clinical challenge. Current methods rely heavily on post-operative pathology, a process that is inherently invasive and offers no opportunity for early treatment adjustment.

Pioneering a Noninvasive Predictive Framework
This latest research, led by a collaborative team from the Jiangsu Key Laboratory of Intelligent Medical Image Computing at Nanjing University of Information Science and Technology and the Department of Nuclear Medicine at Shanghai Proton and Heavy Ion Center, Fudan University Cancer Hospital, has explored the utility of baseline D-PET imaging in conjunction with sophisticated AI algorithms. The study aimed to develop and evaluate models capable of predicting pCR before treatment even begins.
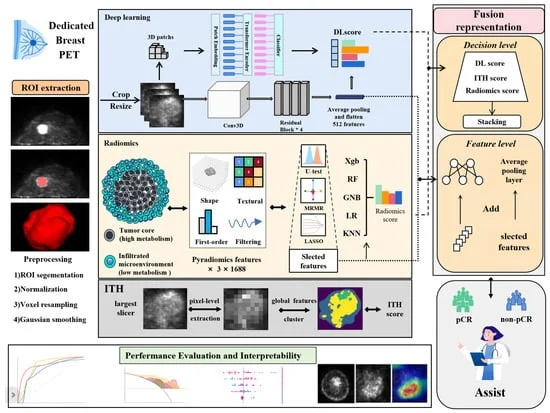
The researchers employed a multi-faceted approach, integrating two powerful AI methodologies: radiomics and deep learning. Radiomics involves extracting a large number of quantitative features from medical images, such as texture, shape, and intensity patterns, which can then be analyzed to identify subtle indicators of disease. Deep learning, on the other hand, utilizes complex neural networks to automatically learn intricate patterns directly from volumetric image data.

Methodology: A Multi-Pronged AI Approach
The study involved a cohort of 147 patients with HER2-positive breast cancer who underwent baseline D-PET imaging prior to commencing neoadjuvant chemotherapy. The research team meticulously designed a framework that analyzed the D-PET scans through three distinct pathways:
- Radiomics: This pathway focused on extracting quantitative features from the entire tumor volume and also from data-driven high- and low-uptake metabolic subregions within the tumor. These subregions were identified using clustering techniques, aiming to capture variations in metabolic activity that might correlate with treatment response.
- Deep Learning: A 3D ResNet convolutional neural network was trained on standardized 3D tumor volumes from the D-PET scans. This approach allowed the AI to automatically learn complex, high-level features indicative of treatment response.
- Intratumoral Heterogeneity (ITH): As a supplementary metric, the researchers quantified intratumoral heterogeneity on the largest slice of the tumor, a measure known to sometimes correlate with treatment outcomes.
Crucially, the study didn’t stop at these individual analyses. To harness the complementary strengths of radiomics and deep learning, the researchers implemented fusion strategies. These strategies aimed to combine the predictive power of both AI approaches, offering a more robust and accurate prediction than either method could achieve alone. Two fusion techniques were explored:

- Feature-Level Fusion: This involved integrating the learned representations from the deep learning model with the handcrafted radiomic features at an intermediate stage of the AI pipeline.
- Decision-Level Fusion: In this approach, the final predictions from the individual radiomics and deep learning models were combined, typically through a secondary learning algorithm, to produce a final, fused prediction.
Robust Results: Fusion Models Excel
The findings from the study demonstrated that both the deep learning and radiomics models, when used individually, achieved comparable and promising performance in predicting pCR on the primary test set, with Area Under the Receiver Operating Characteristic Curve (AUC) values of 0.79 and 0.78, respectively. The ITH model, however, showed limited predictive value.
The true power of the AI framework emerged when the fusion strategies were applied. On the primary test set, feature-level fusion achieved the highest AUC of 0.84, closely followed by decision-level fusion with an AUC of 0.83. This indicated that combining the information from radiomics and deep learning significantly enhanced the models’ ability to discriminate between responders and non-responders.

Further validation on a temporally separated, albeit smaller, test set (Test Set 2) showed consistent trends. Decision-level fusion achieved the highest AUC of 0.84, underscoring the robustness of integrating predictions from different AI approaches.
Interpreting the Findings: Towards Clinical Applicability
Beyond statistical performance, the study placed significant emphasis on model interpretability. Techniques like Gradient-weighted Class Activation Mapping (Grad-CAM) were used to visualize which areas of the D-PET scans the deep learning models were focusing on, revealing that they often highlighted regions with high metabolic activity. SHAP (SHapley Additive exPlanations) values provided insights into the contribution of specific radiomic features and the relative importance of the deep learning and radiomics branches in the fusion models. This transparency is crucial for building trust and facilitating the adoption of AI tools in clinical practice.

The researchers also explored the clinical implications of their findings. They noted that traditional baseline clinical and pathological factors showed limited predictive ability in this HER2-positive cohort, suggesting that advanced imaging biomarkers are essential for accurate response prediction. The study highlighted how such predictive models could potentially aid in surgical planning, allowing for more informed decisions regarding breast-conserving surgery versus mastectomy, and ultimately contributing to a more individualized treatment journey for patients.
Official Responses and Future Directions
The study’s authors, led by Professor Wenlong Ming and Dr. Jingyi Cheng, emphasized the exploratory nature of their findings. "Our results are highly encouraging and suggest that baseline D-PET imaging, when analyzed with sophisticated AI tools, can provide valuable noninvasive insights into treatment response," stated Professor Ming. "The fusion of deep learning and radiomics shows particular promise in enhancing predictive accuracy."

Dr. Cheng added, "While these findings represent a significant step forward, further large-scale, multi-institutional prospective studies are imperative to validate these models, confirm their generalizability across diverse patient populations and imaging protocols, and ultimately integrate them seamlessly into clinical decision-making pathways."
The research team has made their code publicly available to promote reproducibility and further research in this critical area. They are committed to refining these AI models and exploring their application to other breast cancer subtypes and treatment modalities.

Implications for Patient Care
The implications of this research are substantial. By accurately predicting which patients are likely to achieve a complete response to neoadjuvant chemotherapy, clinicians can:
- Optimize Treatment Strategies: Patients predicted to respond well might receive standard therapy, while those less likely to respond could be considered for alternative or intensified treatment regimens earlier in their care.
- Reduce Unnecessary Interventions: Avoiding aggressive chemotherapy for patients who are unlikely to benefit can spare them from debilitating side effects and associated healthcare costs.
- Enhance Surgical Planning: A reliable prediction of tumor shrinkage can inform surgical approaches, potentially increasing the rates of breast-conserving surgery and improving cosmetic outcomes.
- Improve Patient Prognosis: By tailoring treatments more precisely, the ultimate goal is to improve survival rates and quality of life for individuals diagnosed with HER2-positive breast cancer.
This pioneering work underscores the transformative potential of integrating advanced imaging technologies with cutting-edge AI to revolutionize cancer care, moving towards a future where treatment is not only more effective but also more precisely tailored to the individual patient.





