New Delhi, India – In the relentless battle against cancer, scientists are delving deeper into the intricate molecular mechanisms that govern cell survival and death. A critical area of focus is the complex interplay between two fundamental cellular processes: autophagy and apoptosis. While seemingly opposing forces – autophagy promoting survival through cellular recycling, and apoptosis orchestrating programmed cell death – their intricate crosstalk plays a pivotal role in cancer development, progression, and response to treatment. Recent research published in the journal Cancers sheds light on this complex relationship, identifying key molecular players and signaling pathways that could unlock novel therapeutic strategies.

The Delicate Balance: Autophagy and Apoptosis in Cancer
At its core, cellular life and death are determined by a finely tuned balance. Autophagy, a cellular "self-eating" mechanism, degrades damaged organelles and misfolded proteins, acting as a survival strategy under stress. Apoptosis, conversely, is a programmed cell death pathway essential for eliminating damaged or unwanted cells. In the context of cancer, this balance is often disrupted, with tumor cells exploiting autophagy to survive harsh conditions, such as nutrient deprivation and chemotherapy, thereby fostering resistance to treatment.

However, the relationship is far from simple. Under certain circumstances, autophagy can paradoxically trigger cell death, and components of the apoptotic machinery can influence autophagic processes. This complex interplay, known as crosstalk, is mediated by a network of proteins and signaling pathways, making it a prime target for therapeutic intervention.

Unmasking the Molecular Architects: Key Players in the Crosstalk
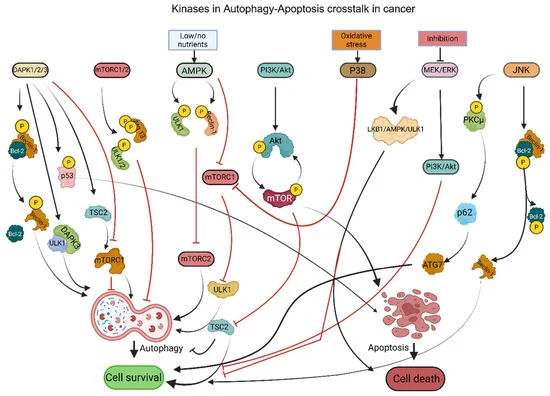
The review highlights a cast of molecular players that orchestrate the autophagy-apoptosis dialogue. Kinases, such as Death-Associated Protein Kinase (DAPK), Mechanistic Target of Rapamycin (mTOR), and AMP-activated Protein Kinase (AMPK), are central regulators. DAPK, for instance, can promote autophagy by releasing Beclin-1 from anti-apoptotic proteins, but can also induce apoptosis. mTOR, a critical nutrient sensor, generally suppresses autophagy, while AMPK activates it during energy deficit, often pushing cells towards death. The Phosphoinositide 3-Kinase (PI3K)/Akt pathway, frequently hyperactivated in cancer, promotes survival by inhibiting apoptosis and autophagy. Stress-activated kinases like p38 MAPK and JNK also play dual roles, their activation under stress potentially leading to either survival via autophagy or programmed death.

Caspases, long recognized as the executioners of apoptosis, are also implicated. Their cleavage of autophagy-related proteins can inhibit autophagy, thereby tipping the balance towards cell death. Conversely, autophagy can regulate caspase activity, for example, by clearing pro-apoptotic caspases. Heat shock proteins (HSPs), particularly HSP70 and HSP90, act as chaperones that can influence cell fate by stabilizing proteins involved in both pathways, often promoting survival by preventing cell death signals.

Transcription factors wield significant control over gene expression, dictating the cellular response to stress. C/EBP Homologous Protein (CHOP) and Activating Transcription Factor 4 (ATF4) are key players in the Unfolded Protein Response (UPR), a pathway activated by endoplasmic reticulum (ER) stress. While UPR can be adaptive, prolonged ER stress mediated by CHOP and ATF4 can drive apoptosis. Forkhead Box O (FOXO) transcription factors, regulated by pathways like PI3K/Akt, can promote autophagy under mild stress and apoptosis under severe stress. Hypoxia-Inducible Factor-1α (HIF-1α) links low oxygen conditions to both autophagy and apoptosis, influencing survival in the tumor microenvironment. Nuclear Factor Erythroid 2-Related Factor 2 (NRF2) regulates antioxidant responses and can influence both autophagy and apoptosis, often contributing to cancer cell resistance. P53, the renowned tumor suppressor, determines cell fate by inducing autophagy under less severe stress and apoptosis when damage is significant. Signal Transducer and Activator of Transcription 3 (STAT3) and Nuclear Factor Kappa B (NF-κB) are crucial in integrating inflammatory and survival signals, often promoting cancer cell survival by modulating both autophagy and apoptosis.

Signaling Pathways: The Communication Network
The intricate dance between autophagy and apoptosis is orchestrated by sophisticated signal transduction pathways. The Unfolded Protein Response (UPR), triggered by ER stress, can initiate adaptive autophagy for survival or, if stress persists, drive apoptosis. Oxidative stress, characterized by reactive oxygen species (ROS), has a dose-dependent effect: low ROS levels can promote proliferation, while high levels can induce apoptosis and autophagy, influencing cancer progression and treatment response. The MAPK pathway (including ERK, JNK, and p38) acts as a critical switch, with different components influencing cell fate decisions by modulating autophagy and apoptosis regulators. Calcium signaling acts as a rheostat, with alterations in intracellular calcium levels dictating whether a cell opts for autophagic survival or apoptotic commitment.

Therapeutic Horizons: Natural Products and FDA-Approved Drugs
Understanding this complex crosstalk opens exciting avenues for cancer therapy. The review highlights the potential of natural compounds derived from plants, which have long been recognized for their therapeutic properties. Compounds like curcumin, berberine, resveratrol, and quercetin have shown promise in modulating the autophagy-apoptosis balance. They can trigger apoptosis through various mechanisms, including mitochondrial dysfunction and ER stress, while also influencing autophagic flux. These phytochemicals can inhibit pro-survival autophagy or activate cytotoxic forms, offering a complementary approach to traditional chemotherapy with potentially lower toxicity.

Furthermore, FDA-approved drugs, initially developed for other conditions, are being repurposed for their ability to modulate this critical cellular dialogue. Drugs like chloroquine and hydroxychloroquine, known for their antimalarial properties, inhibit the late stages of autophagy, thereby enhancing the apoptotic effects of chemotherapy. Metformin, a diabetes drug, has shown potential in sensitizing cancer cells to apoptosis by activating AMPK and inhibiting mTOR. Other approved agents, including sorafenib, everolimus, and bortezomib, are also being investigated for their roles in modulating autophagy-apoptosis crosstalk, often in combination therapies to overcome drug resistance and improve treatment efficacy.

Implications for Cancer Treatment
The intricate relationship between autophagy and apoptosis presents a formidable challenge but also a significant opportunity in the fight against cancer. By understanding the molecular mechanisms that govern this balance, researchers aim to develop targeted therapies that can precisely manipulate these pathways. This could involve:

- Inhibiting pro-survival autophagy: Blocking the autophagic machinery that cancer cells exploit to survive chemotherapy.
- Activating cytotoxic autophagy: Inducing autophagic processes that lead to cell death.
- Synergistic drug combinations: Combining drugs that target autophagy and apoptosis pathways to achieve a more potent and selective killing of cancer cells.
- Leveraging natural compounds: Harnessing the multitargeted effects of phytochemicals to overcome drug resistance and improve therapeutic outcomes.
The ongoing research into autophagy-apoptosis crosstalk underscores a paradigm shift in cancer therapy, moving towards a more personalized and nuanced approach that targets the fundamental cellular processes driving tumor survival and death. As our understanding deepens, the prospect of developing novel, effective treatments that can overcome therapeutic resistance and improve patient survival becomes increasingly tangible.




