The rapid ascent of Medvi, a digital health startup recently lauded by The New York Times as a "vibe-coded unicorn," has been met with a jarring reality check. While CEO Matthew Gallagher’s venture has successfully captivated the tech-bro zeitgeist, a growing body of investigative reporting suggests the company’s business model is built on a foundation of regulatory skirting, aggressive digital marketing, and questionable medical practices.
Beyond the well-documented controversy surrounding the company’s compounding of GLP-1 weight-loss drugs—which has already drawn a formal FDA warning letter—a more alarming concern has emerged: QUAD. This is a compounded erectile dysfunction (ED) formula marketed by Medvi that crams four powerful pharmacological agents into a single sublingual dose. The product is not FDA-approved, and medical experts are sounding the alarm over its potential for dangerous cardiovascular side effects.
The Anatomy of QUAD: A Clinical Minefield
QUAD is marketed as a "Complete Stack" for erectile dysfunction, combining three distinct PDE5 inhibitors—sildenafil (Viagra), tadalafil (Cialis), and vardenafil (Levitra)—with apomorphine, a drug typically reserved for managing the motor fluctuations of advanced Parkinson’s disease.

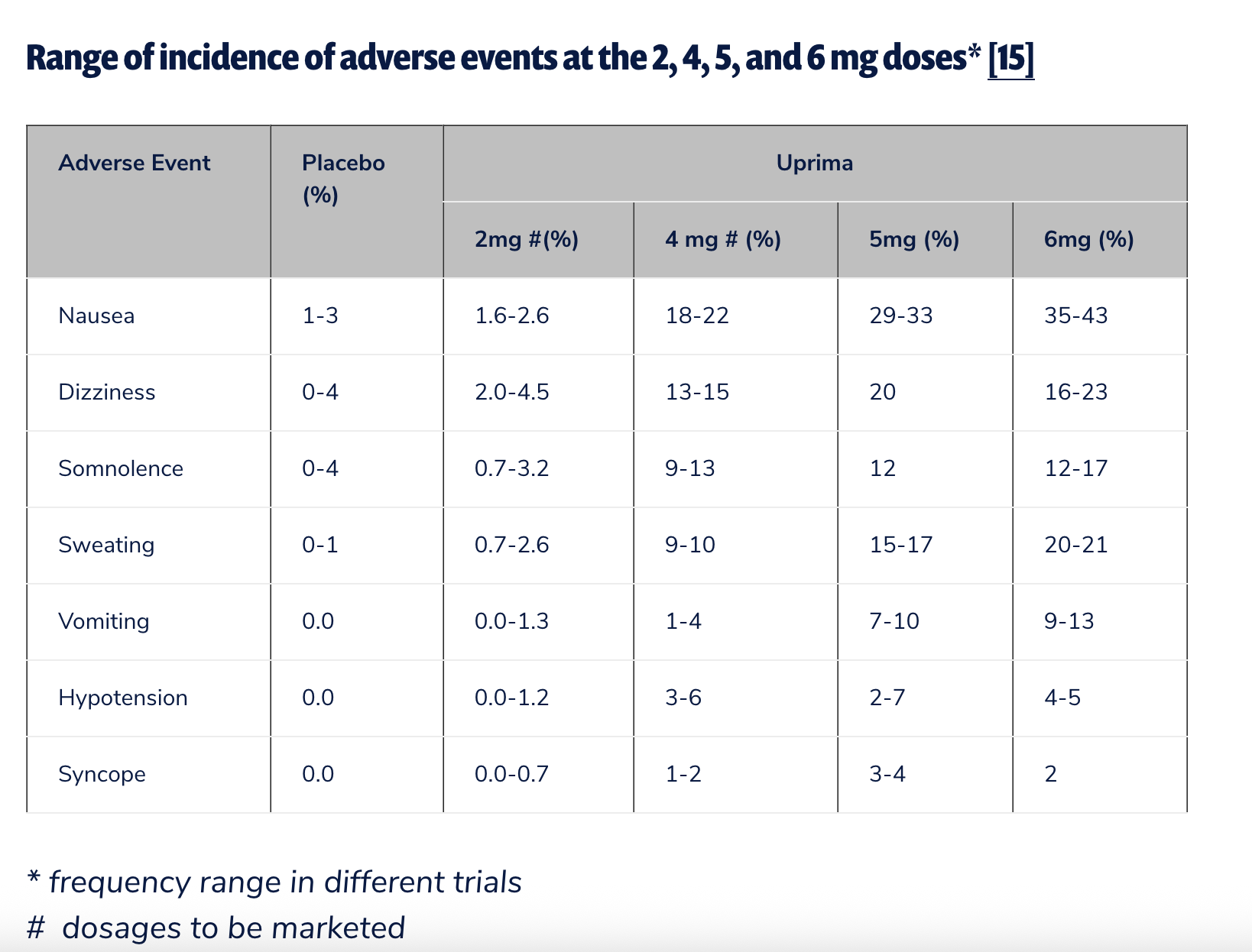
The inclusion of apomorphine is particularly contentious. In the United States, apomorphine is not FDA-approved for the treatment of erectile dysfunction. Its primary use in human medicine is for Parkinson’s, and in veterinary medicine, it is frequently used to induce vomiting in dogs. While the drug was explored for ED in Europe under the brand name Uprima, its marketing authorization expired in 2006. Many patients stopped using it due to a high incidence of nausea and a general lack of efficacy.
Most concerning to pharmacologists is the "stacking" of these four agents. The FDA has historically warned that the combination of multiple PDE5 inhibitors has not been rigorously studied, carries an increased risk of severe, sustained hypotension (low blood pressure), and is generally not recommended by clinical guidelines. Professional drug-interaction databases, such as Drugs.com, categorize the combination of these agents as a "Major" interaction, warning of risks ranging from fainting to cardiac events.
Chronology of a Regulatory Storm
The scrutiny of Medvi did not happen in a vacuum. It is the culmination of months of aggressive expansion and subsequent regulatory pushback.
- June 2000: The medical community long anticipated the risks of apomorphine for ED. Public Citizen’s Sidney Wolfe, M.D., famously urged the FDA to reject the drug, warning that its approval would lead to "serious injuries and a near-certain post-market ban."
- February 2026: The FDA issued a formal warning letter to Medvi, LLC, citing the company for the misbranding of compounded GLP-1 drugs.
- April 2, 2026: The New York Times published a profile of CEO Matthew Gallagher, framing his company as a revolutionary force in digital health, sparking intense debate regarding the role of AI-driven startups in clinical spaces.
- April 7, 2026: Investigative testing conducted by Drug Discovery & Development revealed significant flaws in the company’s automated intake software, which appeared to bypass critical safety screenings for patients with pre-existing cardiovascular conditions.
- Present: Medvi faces a growing list of lawsuits and ongoing scrutiny from health regulators and federal investigators.
Digital Marketing vs. Patient Safety
Medvi’s growth strategy relies heavily on digital customer acquisition through affiliate marketing networks. These platforms often use slick, AI-generated "doctor" personas and generic stock imagery to create a veneer of professional endorsement.
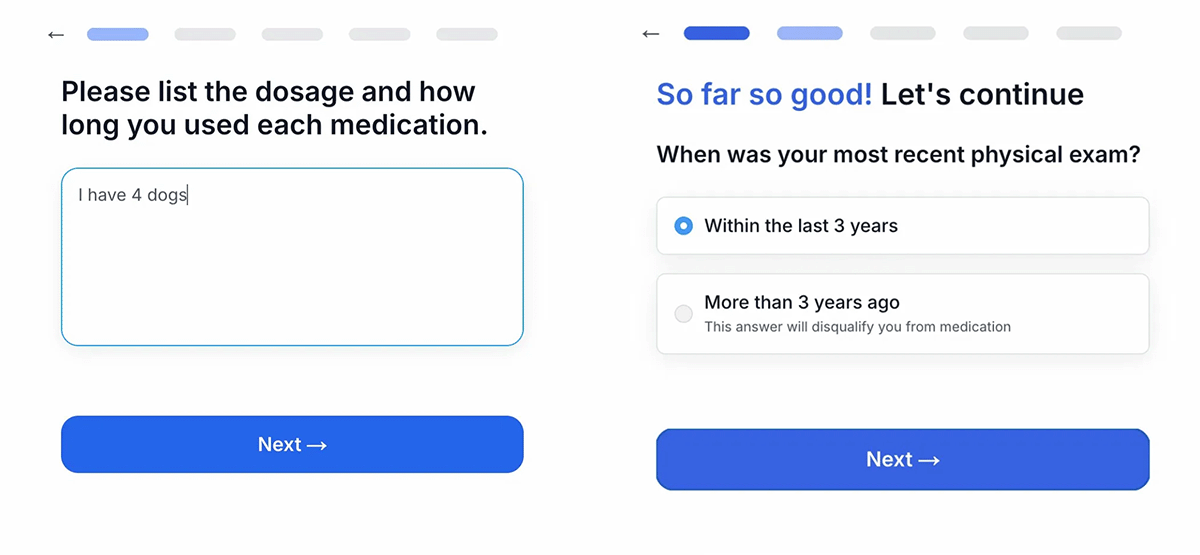
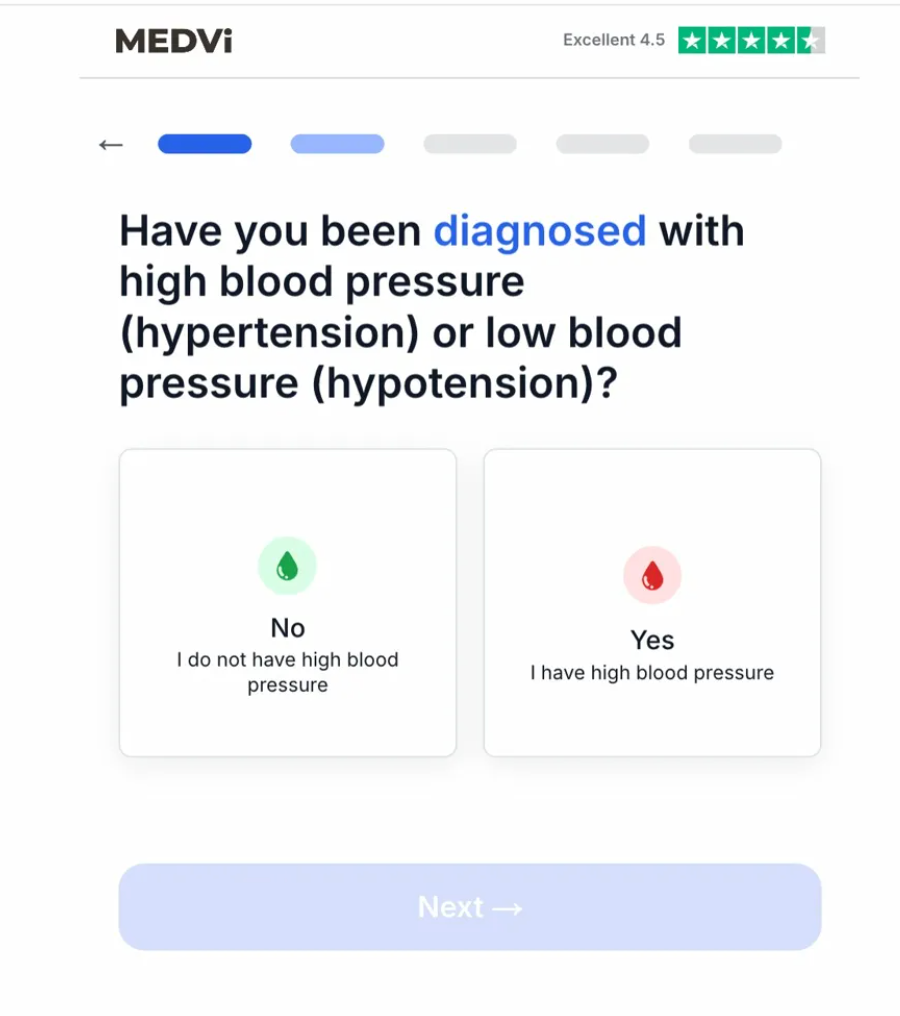
During April 2026 testing, the onboarding portal for QUAD was found to have alarming technical oversights. In one instance, the intake tool asked users if they had been diagnosed with hypotension (low blood pressure), yet provided no mechanism to flag or screen out those who answered "yes." In another test, the system appeared to ignore free-form text responses that indicated disqualifying medical histories, funneling users toward a prescription regardless of the input.
Furthermore, the "94% success probability" rate cited on the Medvi onboarding screen—a statistic that conveniently matches the figure used for their GLP-1 weight-loss products—lacks transparent clinical backing. This number appears in various marketing materials, serving more as a psychological conversion tactic than a reflection of validated clinical data.
The "Essentially a Copy" Regulatory Loophole
The FDA’s current stance on compounding provides a narrow pathway for pharmacies to prepare individualized medicines for patients who cannot use commercially available products. However, the agency’s April 1, 2026, guidance clarifies that a compounded product is considered "essentially a copy" if it contains the same active ingredients as commercially available products.
While the FDA allows for small-batch exceptions (four or fewer prescriptions per month), Medvi’s business model appears designed to operate at a scale that challenges this definition. By combining four distinct drugs into a single sublingual dose, Medvi is effectively creating a new product that has never undergone the rigorous, multi-year clinical trials required for FDA approval.
Official Responses and Industry Accountability
When asked about the safety and compliance of the QUAD product, the U.S. Department of Health and Human Services (HHS) declined to comment on specific company matters. A spokesperson noted, "The FDA generally does not discuss compliance matters except with the company involved," while reiterating that the agency takes appropriate action when violations occur.
The industry at large has been slower to react, but the legal community is circling. In Day v. OpenLoop Health, a federal class-action lawsuit currently pending in Delaware, the spotlight is turning toward the intersection of telehealth platforms and the pharmacy networks that fill their prescriptions. While Medvi is not the lead defendant, its presence in the case underscores the growing liability for companies that prioritize rapid growth over medical due diligence.
Implications for the Future of Telehealth
The Medvi saga raises existential questions for the booming "DTC" (direct-to-consumer) medical industry. The promise of the digital revolution was the democratization of healthcare—easier access, lower costs, and more personalized treatments. However, the use of AI to automate medical screening and the aggressive marketing of unapproved, compounded "cocktails" suggests a darker potential: the normalization of medical risk in the name of speed and scale.
For patients, the implications are profound. When an online portal promises a "quick fix" for complex conditions like erectile dysfunction, the line between helpful innovation and medical malpractice blurs. The case of QUAD serves as a warning that without rigorous oversight, the "vibe-coded" solutions of today may become the public health liabilities of tomorrow.

As federal agencies tighten their grip on compounding practices, startups like Medvi will likely be forced to pivot or face existential legal challenges. Whether they can reconcile their "move-fast" Silicon Valley ethos with the "do-no-harm" mandate of the medical profession remains to be seen. For now, the medical community remains vigilant, watching to see if the FDA’s warnings translate into meaningful enforcement or if, like many tech-disruptors before them, Medvi will continue to operate in the gray zones of regulation until a tragedy forces their hand.