By Scientific Correspondence Desk
For over seven decades, S-methylmethionine (SMM)—widely known in historical medical texts as "Vitamin U"—has existed in a state of scientific limbo. First identified in the 1940s for its remarkable ability to support gastrointestinal health, the compound has remained a staple of folk medicine and niche nutraceutical markets. However, a comprehensive new review published in the journal Pharmaceuticals (Vol. 19, No. 5, 2026) suggests that the time has come to re-evaluate this sulfur-containing compound through the lens of modern molecular biology.
The study, led by Arsen A. Ananian and a multi-disciplinary team from Sechenov University, provides a rigorous, critical synthesis of SMM’s pharmacological profile. It highlights a disconnect between the compound’s substantial preclinical promise and its relative absence from mainstream clinical practice, ultimately calling for a paradigm shift in how we approach its development.
Main Facts: What is Vitamin U?
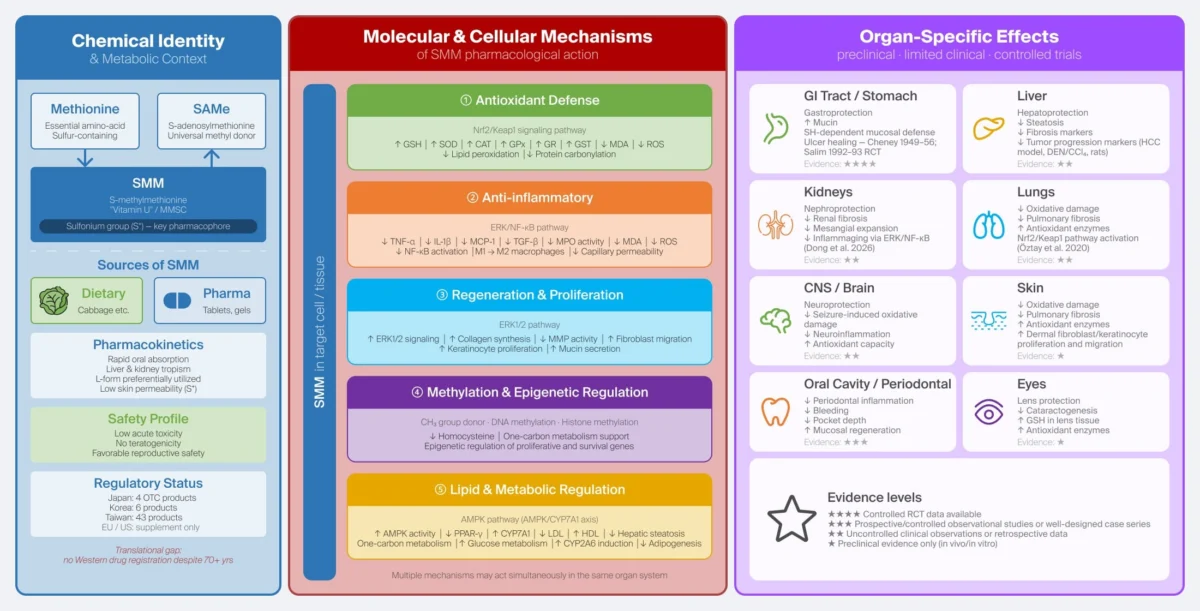
S-methylmethionine is a naturally occurring, sulfur-containing compound found in various plant-based foods. It gained the colloquial moniker "Vitamin U" (the "U" standing for ulcus, the Latin word for ulcer) due to its observed anti-ulcerogenic properties in early 20th-century studies.
At its core, SMM functions as a vital methyl donor in biological systems. While it shares structural similarities with S-adenosylmethionine (SAMe)—a well-known, clinically validated methyl donor used in treating depression and osteoarthritis—SMM possesses unique pharmacokinetic properties that have long intrigued researchers.
The 2026 review synthesizes decades of data, identifying that SMM’s therapeutic potential extends far beyond simple stomach lining protection. Its primary mechanisms of action include:
- Modulation of Oxidative Stress: SMM acts as a potent scavenger of reactive oxygen species (ROS).
- Anti-inflammatory Signaling: It interacts with critical pathways such as the ERK/NF-κB and Nrf2/Keap1 signaling axes, which are essential for cellular defense and the regulation of inflammation.
- Homeostasis Maintenance: By supporting glutathione homeostasis, SMM assists the body in maintaining its primary intracellular antioxidant, thereby protecting cells from damage across diverse organ systems.
A Chronology of Investigation
The history of Vitamin U is one of early enthusiasm followed by long-term stagnation.

- 1940s–1950s: The "Golden Era" of Vitamin U discovery. Researchers observed that extracts from raw cabbage juice facilitated the healing of peptic ulcers, leading to the isolation of SMM.
- 1960s–1980s: Preclinical studies began to expand the scope of SMM research. Scientists observed hepatoprotective, nephroprotective, and cytoprotective effects in animal models. Despite these findings, large-scale human trials failed to materialize in the West.
- 1990s–2010s: The compound was largely relegated to the "alternative medicine" category. While studies continued, primarily in Eastern Europe and Russia, the lack of standardized, high-quality human trials kept SMM off the radar of major pharmaceutical regulators like the FDA or EMA.
- 2026: The publication of the Sechenov University review marks a pivotal moment. By applying modern evidence-grading standards to 70 years of disparate data, the authors aim to bridge the gap between historical observations and contemporary evidence-based medicine.
Supporting Data: Organ-Specific Protection
The review provides a deep dive into the multi-organ benefits of SMM, noting that while human clinical data remains scarce, the preclinical evidence is robust and consistent.
The Gastrointestinal Tract
The most validated indication for SMM remains gastroprotection. The compound appears to accelerate the repair of the gastric mucosa by inhibiting excessive acid secretion and stimulating the regenerative processes of epithelial cells.
Neurological and Systemic Effects
Beyond the gut, the review highlights emerging evidence for neuroprotection. In various models, SMM has been shown to stabilize cellular membranes and reduce inflammatory responses in neural tissue. This opens the door to potential applications in treating neurodegenerative processes where oxidative stress is a primary driver.
Liver and Kidney Health
The liver and kidneys are the primary organs for detoxification and metabolic processing. The study highlights that SMM supports the structural integrity of these organs by optimizing methylation pathways, which are critical for the detoxification of toxins and the regulation of gene expression.
Dermatological and Ocular Applications
Perhaps the most innovative area discussed is the use of SMM in topical and mucosal delivery systems. Given the compound’s ability to promote tissue regeneration, researchers believe there is significant potential for SMM-based formulations in treating chronic wounds, ocular surface inflammation, and oral lesions.
The Translational and Regulatory Barrier
Why, after 70 years of research, is Vitamin U not a standard drug on pharmacy shelves? The authors of the review identify three critical "translational barriers":
- The "Nutraceutical" Stigma: Because SMM is naturally occurring and has been marketed as a supplement for decades, there is little financial incentive for pharmaceutical companies to invest in the multi-million-dollar Phase III trials required for drug registration.
- Lack of Standardization: Historical studies often used varying purities and delivery methods. Modern regulatory bodies require highly standardized, stable pharmaceutical formulations, which were not a priority in early 20th-century research.
- Fragmented Clinical Evidence: While there is a mountain of preclinical data, the "human evidence" remains largely descriptive or anecdotal. The medical community requires well-designed, placebo-controlled Phase II trials to definitively prove efficacy in human populations.
Implications: A Call for Innovative Research
The authors conclude that the future of SMM lies in moving away from generic supplementation and toward targeted pharmaceutical development.

The Need for Phase II Trials
The most urgent recommendation is the initiation of Phase II randomized controlled trials (RCTs). These trials should focus on specific indications where preclinical evidence is strongest, such as the treatment of refractory gastric ulcers or as an adjunct therapy for inflammatory conditions.
Innovative Delivery Systems
The review emphasizes that oral bioavailability is not the only path forward. The development of sophisticated topical gels, mucosal sprays, and controlled-release matrices could allow SMM to be delivered directly to the site of injury, maximizing efficacy while minimizing systemic side effects.
A New Regulatory Path
For SMM to achieve the status it deserves, researchers must engage with regulatory agencies to define a path that acknowledges its long history of safe use while demanding the rigor of modern clinical science.
The Sechenov University team’s work acts as a roadmap for this transition. By systematically stripping away the historical "superfood" narrative and replacing it with a rigorous, evidence-based assessment, the researchers have set the stage for a potential medical renaissance.
As we move toward the latter half of the 2020s, the "Vitamin U" of the past may well become a key therapeutic agent of the future. The challenge now rests with the global scientific community to turn these promising molecular mechanisms into tangible clinical outcomes for patients worldwide.
Source Reference:
Ananian, A.A.; Zelenina, T.Z.; Stepanova, O.I.; Popova, A.A.; Bagatelia, Z.T.; Kosenkova, S.I.; Evzikov, G.Y.; Sysuev, B.B.; Brkich, G.E.; Pyatigorskaya, N.V.; et al. S-Methylmethionine (Vitamin U): A Critical Narrative Review of Pharmacological Mechanisms, Evidence Levels, and Translational Barriers. Pharmaceuticals 2026, 19, 743. https://doi.org/10.3390/ph19050743