Memphis, TN – In a groundbreaking study poised to redefine long-term care for childhood cancer survivors, scientists at St. Jude Children’s Research Hospital have revealed a complex interplay between an individual’s genetic makeup and their life-saving cancer treatments in determining the risk of developing secondary cancers. Published today in The Lancet Oncology, this pivotal research for the first time quantifies the proportional contributions of these factors, challenging conventional wisdom and offering a clearer roadmap for personalized medical surveillance.
Main Facts
For millions worldwide who have triumphed over childhood cancer, the specter of a secondary malignancy looms as the primary cause of mortality in their later lives. Until now, the precise extent to which different factors contribute to this risk has remained largely unquantified at a population level. The new St. Jude study meticulously dissects these contributions, demonstrating unequivocally that a survivor’s inherent genetic predisposition, alongside the therapeutic interventions they received, are the dominant forces shaping their susceptibility to new cancers.
This landmark investigation leveraged an unparalleled dataset drawn from two of the world’s most comprehensive and respected childhood cancer survivor cohorts: the St. Jude Lifetime Cohort Study (St. Jude LIFE) and the Childhood Cancer Survivor Study (CCSS). Housed at St. Jude, these studies collectively represent an invaluable repository of clinical, genetic, and lifestyle data from thousands of survivors, making such an in-depth analysis possible.
"We found the burden of second cancer in survivors of childhood cancer is largely contributed by pediatric treatment exposures and genetic predisposition," stated corresponding author Yadav Sapkota, PhD, from St. Jude’s Department of Epidemiology and Cancer Control. Dr. Sapkota emphasized the novelty of the quantification: "We’ve known treatment exposures and genetics were associated with second cancer risk, but this is the first time we’ve been able to attribute the proportion of their contributions to that risk at the population level." This quantification marks a crucial turning point, providing clinicians and survivors with actionable insights that could fundamentally alter surveillance strategies and empower individuals to better understand their unique risk profiles. The findings underscore a critical message for physicians: care plans for these survivors must integrate an awareness of both their treatment history and their genetic heritage.
Chronology: An Evolving Understanding of Survivorship Risk
The journey to understand and mitigate the long-term health consequences of childhood cancer treatment has been a progressive one, marked by continuous research and evolving clinical practices. In the early days of pediatric oncology, the primary focus was on achieving survival, often at significant cost to future health. As survival rates dramatically improved over the latter half of the 20th century, the medical community began to grapple with the emerging challenges faced by these long-term survivors, among the most serious being the development of secondary cancers.
Initially, research efforts predominantly centered on identifying specific treatment exposures, particularly radiation therapy and certain chemotherapeutic agents, as direct culprits in increasing secondary cancer risk. Seminal studies meticulously documented the adverse effects of radiation, linking it to various solid tumors, and chemotherapy to specific leukemias and lymphomas, as well as solid tumors. This knowledge spurred critical advancements in treatment protocols, leading to the judicious reduction of radiation doses, the development of more targeted chemotherapy regimens, and, in many cases, the complete removal of radiation from treatment plans as other therapies became more effective and less toxic. These proactive adaptations were born from a growing understanding of the dose-dependent and cumulative nature of treatment-related toxicities.
However, even as treatment protocols became safer, secondary cancers persisted, suggesting other factors were at play. The concept of individual susceptibility began to gain traction, leading researchers to consider the role of genetics. Early investigations hinted at familial predispositions and the involvement of specific germline mutations in increasing risk. Yet, these genetic insights often remained siloed, examined in isolation rather than in concert with treatment exposures. The precise interplay—how much of the risk was attributable to the potent effects of therapy versus an individual’s intrinsic genetic vulnerability—remained an unanswered question, representing a significant knowledge gap in the field of survivorship research.
This latest study by St. Jude scientists directly addresses this gap. It represents a culmination of decades of research, moving beyond mere association to concrete attribution. By bringing together the vast, detailed data from the St. Jude LIFE and CCSS cohorts, researchers could for the first time quantitatively dissect the relative contributions of these multifaceted risk factors. This approach not only validates earlier understandings regarding the profound impact of radiation but also introduces a refined perspective on the often-underestimated role of polygenic risk, placing it in direct comparison with the effects of chemotherapy. The study thus marks a critical advancement in the chronological development of survivorship science, shifting the paradigm from identifying risk factors to precisely weighing their individual and collective impact.
Supporting Data: Dissecting the Proportional Contributions to Second Cancers
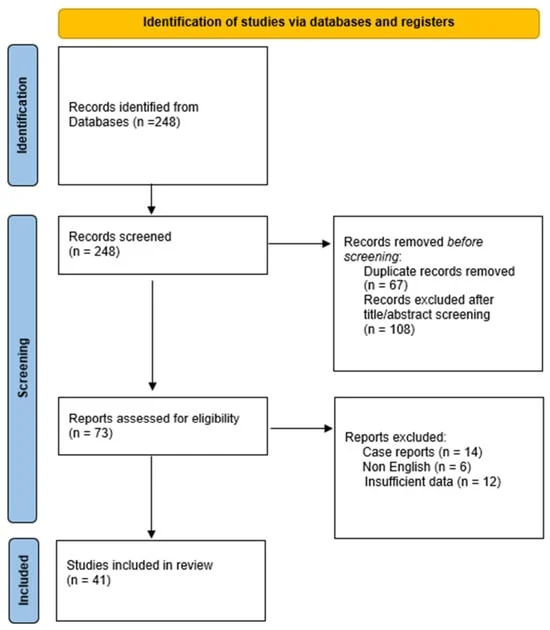
To address the longstanding knowledge gap regarding the relative contributions of various factors to secondary cancer risk, St. Jude scientists undertook an ambitious comparative analysis. Their methodology involved examining an unprecedented cohort of over 10,000 survivors drawn from the St. Jude LIFE and CCSS studies, which, combined, constitute the largest survivor cohort in North America with extensive genetic sequencing data. This robust dataset allowed for a granular evaluation of how distinct elements – ranging from intensive medical treatments to inherited predispositions and lifestyle choices – collectively shape a survivor’s long-term health trajectory.
The researchers meticulously gathered and analyzed a wealth of information for each participant. This included detailed records of their original cancer diagnoses and the specific treatment exposures received, such as cumulative doses of radiation therapy and various chemotherapeutic agents. Alongside this, comprehensive genetic information was compiled, encompassing both hundreds of common genetic variants (aggregated into a polygenic risk score) and rare genetic variants known to be associated with cancer development in the general population. Lifestyle factors, such as diet and exercise habits, were also incorporated, as was the critical outcome measure: the presence or absence of a second primary cancer. This holistic approach provided the empirical foundation necessary to disentangle and quantify the individual and interactive effects of these complex variables.
The findings yielded striking numerical clarity:
-
Radiation Exposure: Emerged as the most significant individual contributor to secondary cancer risk, accounting for approximately 40% or more of the overall risk. This finding strongly reinforces prior research on the long-term adverse effects of radiation, providing compelling data that further supports ongoing efforts in modern oncology to lower radiation doses or eliminate its use entirely when alternative effective treatments are available. The cellular damage induced by radiation is a potent carcinogenic trigger, and its substantial contribution underscores the success and necessity of these evolving treatment paradigms.
-
Chemotherapy: The impact of various chemotherapeutic agents was found to be more variable, contributing between 8% and 35% of subsequent cancer risk, depending on the specific cancer type. While the late effects of chemotherapy, including secondary malignancies, have been well-documented, the precise range of its contribution had not been so clearly delineated at a population level. This variability highlights the diverse mechanisms of action and long-term toxicities associated with different chemotherapy drugs.
-
Genetic Predisposition (Polygenic Risk Score): Perhaps one of the most revelatory findings concerned the contribution of genetics. The researchers utilized a polygenic risk score (PRS), which aggregates the effects of numerous common genetic variants, each individually conferring a small increase in cancer risk. This innovative approach revealed that, depending on the cancer type, the polygenic risk score contributed a substantial 5% to 37% of the secondary cancer risk. This range is particularly significant as it places genetic predisposition on par with, and in some instances, even surpassing, the contribution of chemotherapy. The study also considered rare genetic variants, which, while less common, can confer a very high individual risk. The inclusion of both common and rare variants provides a comprehensive picture of inherited susceptibility.
-
Lifestyle Factors: In contrast to the other factors, lifestyle elements such as diet and exercise appeared to contribute much less, accounting for a modest 1% to 6% of second cancer risk within this cohort. However, the researchers offered a crucial caveat: the majority of survivors in this study were relatively young, primarily in their 20s and 30s. It is plausible that the cumulative effects of lifestyle choices on cancer risk may not yet have had sufficient time to manifest within this younger demographic, suggesting their impact might become more pronounced in older survivor populations.
"This kind of high-impact discovery is only possible in the CCSS and SJLIFE cohorts, that in combination, have more than 12,000 survivors with genetic sequencing," remarked co-author Greg Armstrong, MD, MSCE, chair of the St. Jude Department of Epidemiology and Cancer Control, underscoring the unique power of these expansive datasets. The robust statistical power afforded by such a large, well-characterized cohort allowed the researchers to tease apart these complex relationships with unprecedented clarity and precision, providing a foundational understanding for future clinical applications.
Official Responses: Challenging Conventional Wisdom and Shaping Future Care
The findings from St. Jude have elicited significant responses from the research team, highlighting the transformative potential of this study for both clinical practice and the broader understanding of cancer survivorship. The quantification of genetic and treatment contributions is seen as a pivotal step, moving the field beyond general associations to precise, actionable data.
Dr. Yadav Sapkota, the corresponding author, underscored the study’s challenge to long-held assumptions within the oncology community. "Our findings showed that genetics can be equally or more important than chemotherapy in some second cancers, which is counter to conventional wisdom in the field," Sapkota stated. This revelation is particularly impactful because, for decades, the focus in assessing secondary cancer risk has predominantly been on charting treatment exposures, with genetic predisposition often playing a secondary, less quantified role. The data now demands a re-evaluation of this hierarchy, suggesting that an individual’s inherited susceptibility can be a formidable, sometimes even dominant, force.
Co-author Dr. Yutaka Yasui, also from St. Jude’s Department of Epidemiology and Cancer Control, elaborated on the clinical utility of the advanced genetic tools employed in the study. "Polygenic risk scores are developed for all kinds of diseases for personalized medicine, but generally with precision below what is required for clinical utility in the general population," Yasui explained. However, he quickly pivoted to the specific context of childhood cancer survivors: "Among survivors of childhood cancer and for estimating their risk of certain types of subsequent cancer, however, they may provide useful information in conjunction with therapy exposures." This distinction is critical; while PRSs might not yet be universally precise enough for broad clinical application, their utility is significantly amplified in a population already at elevated risk due to specific exposures, making them a powerful adjunct to traditional risk assessment.
Regarding lifestyle factors, Dr. Sapkota provided important nuance to their seemingly minor contribution in this study. "We know healthy lifestyle choices are important for survivors," he affirmed. "In this study, we focused only on the risk of second cancers, which may not be strongly impacted by lifestyle at this young age. However, other research has shown the benefits of healthy choices on other late effects, such as protecting cardiac wellbeing, so it is still important for clinicians to encourage — and patients to seek — a healthy lifestyle." This clarification ensures that the findings on lifestyle are not misinterpreted as diminishing the overall importance of healthy living for survivors, but rather as contextualizing its specific impact on secondary cancer risk within a younger age group.
The consensus among the researchers is clear: this study represents a significant leap forward in understanding the multifactorial nature of secondary cancer risk. It provides the empirical evidence necessary to advocate for a more comprehensive and personalized approach to survivorship care, moving beyond a sole reliance on treatment history to integrate the powerful insights offered by an individual’s genetic blueprint. These official responses reflect a commitment to translating cutting-edge research into tangible improvements in patient care, ultimately aiming to enhance the quality and longevity of life for childhood cancer survivors.
Implications: Reshaping Survivorship Care for a Healthier Future
The insights gleaned from this landmark St. Jude study carry profound implications for the future of childhood cancer survivorship, promising to reshape clinical paradigms, empower patients, and guide future research efforts. By quantifying the proportional contributions of treatment exposures and genetic predisposition, the research offers a clearer, more nuanced understanding of secondary cancer risk, moving the field towards truly personalized medicine.
Shifting Clinical Paradigms
The most immediate and impactful implication is the necessity for a fundamental shift in how physicians assess and manage secondary cancer risk in survivors. As Dr. Sapkota highlighted, "Historically, we have paid attention to survivors’ treatment exposures when determining second cancer risk. Our study suggests that we need to better account for genetic predisposition in this population." This calls for the integration of genetic screening, particularly polygenic risk scores and analysis of rare variants, into routine long-term follow-up care.
For clinicians, this means moving beyond a checklist of past treatments to incorporate a comprehensive genetic profile. Survivors identified with a strong genetic predisposition, either through high polygenic risk scores or specific rare variants, could then be placed on more intensified surveillance protocols. This might include more frequent imaging, earlier initiation of screening tests (e.g., mammograms, colonoscopies), or specialized examinations tailored to the specific cancers to which they are genetically predisposed. The goal is proactive intervention: catching a second cancer at its earliest, most treatable stage, thereby significantly improving prognosis and survival rates. This tailored approach moves away from a "one-size-fits-all" model, recognizing the unique risk landscape of each survivor.
Empowering Patients Through Knowledge
Beyond the clinical sphere, this research empowers survivors with invaluable knowledge about their individual risk factors. Understanding their unique combination of treatment-related, genetic, and even lifestyle contributions to secondary cancer risk allows them to become more informed and proactive advocates for their own health.
Armed with this data, survivors can engage in more meaningful discussions with their healthcare providers, asking pointed questions about personalized screening recommendations and risk reduction strategies. This shift from passive recipient of care to active participant can foster greater adherence to surveillance guidelines and a deeper sense of control over their long-term health. Knowing their genetic predispositions might also influence personal health decisions, encouraging heightened vigilance and a commitment to healthy behaviors, even if the direct impact of lifestyle on secondary cancer risk appears modest in younger cohorts.
Guiding Future Research and Prevention Strategies
The study also lays a robust foundation for future research. While it quantified contributions, it opens new avenues for exploring the precise molecular mechanisms by which specific genetic variants interact with different treatment exposures to drive carcinogenesis. Future studies could focus on:
- Refining Polygenic Risk Scores: Developing even more precise and clinically actionable PRSs for various secondary cancer types, specifically optimized for the unique biological context of childhood cancer survivors.
- Investigating Gene-Environment Interactions: Delving deeper into how specific genetic predispositions are amplified or mitigated by particular chemotherapy drugs or radiation fields.
- Long-term Lifestyle Impact: Conducting longer-term studies on older survivor cohorts to fully understand the cumulative effect of lifestyle factors on secondary cancer risk across the lifespan.
- Developing Targeted Prevention: Identifying high-risk genetic profiles could lead to the development of chemopreventive agents or targeted interventions for specific survivor subgroups.
Ultimately, the insights gained from this research are crucial for developing more effective prevention strategies and improving early detection methods. By understanding where the risk originates, researchers and clinicians can focus their efforts more efficiently, aiming to reduce the incidence and mortality of secondary cancers.
Policy and Resource Allocation
Finally, these findings carry implications for healthcare policy and resource allocation. Implementing widespread genetic screening and personalized surveillance programs will require significant investment in infrastructure, training for healthcare professionals, and accessible genetic counseling services. Policymakers and healthcare systems will need to adapt to support these evolving standards of care, ensuring that all childhood cancer survivors have equitable access to the advanced diagnostics and personalized management strategies that this research now makes possible.
As Dr. Sapkota profoundly concluded, "Second cancers remain the leading cause of mortality for childhood cancer survivors. Now that we have quantified the contributions of treatment, genetics and lifestyle to the risk of secondary disease, we have a better understanding of where to focus efforts to prevent, detect and treat these cancers, and hopefully extend these survivors’ lives." This study is not just an academic achievement; it is a beacon of hope, promising a future where survivors can live longer, healthier lives, free from the shadow of subsequent malignancies.
Authors and Funding
The study’s first author is Achal Neupane, of St. Jude. The study’s other authors are Siddhant Taneja, Jennifer French, Matthew Ehrhardt, Tara Brinkman, Rachel Webster, Jun Yang, Kirsten Ness, Melissa Hudson, Gregory Armstrong, Leslie Robison and Yutaka Yasui; St. Jude; Qi Liu; University of Alberta; Cindy Im, Lucie Turcotte and Joseph Neglia; University of Minnesota; Monica Gramatges, Baylor College of Medicine; Rebecca Howell, University of Texas MD Anderson Cancer Center and Smita Bhatia; University of Alabama at Birmingham.
The study was supported by grants from the National Cancer Institute (R01HL173881, R01CA216354, R21CA261833, U24CA55727, U01CA195547 and CA21765) and ALSAC, the fundraising and awareness organization of St. Jude.