As Medicaid spending on prescription drugs continues to climb, the federal government and state legislatures are intensifying efforts to rein in costs. While pharmaceuticals represent a relatively small portion of total Medicaid expenditures, their rapid price growth has turned drug affordability into a central policy battleground. In response, the Trump administration has launched the GENEROUS (GENErating cost Reductions fOr U.S. Medicaid) Model—a bold, voluntary initiative designed to align domestic drug prices with those paid in other developed nations.
The core of this strategy rests on the "Most-Favored-Nation" (MFN) principle: the belief that the U.S. should not pay higher prices for life-saving medications than comparable, wealthy nations. While the potential for savings is significant, the actual impact of the GENEROUS model remains shrouded in complexity, influenced by confidential industry agreements, shifting tariff policies, and the intricate web of existing state-level rebate programs.
Main Facts: How the GENEROUS Model Operates
The GENEROUS model, launched by the Centers for Medicare & Medicaid Services (CMS) in January 2026, seeks to leverage international pricing data to lower the net cost of brand-name drugs in the Medicaid program.
The Mechanism of Price Alignment
For drugs included in the model, CMS calculates an "MFN price." This is derived from the second-lowest net price reported by manufacturers across eight reference countries: the United Kingdom, France, Germany, Italy, Canada, Japan, Denmark, and Switzerland. To ensure comparability, this figure is adjusted for gross domestic product (GDP) per capita using purchasing power parity. Once the MFN price is determined, CMS negotiates supplemental rebates with participating manufacturers to ensure the Medicaid net price is brought down to that international benchmark.
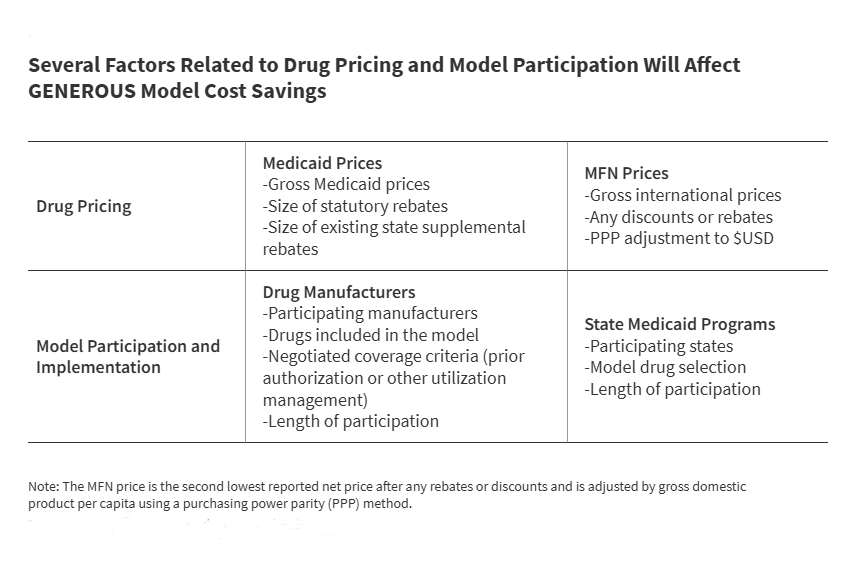
Voluntary Participation
The model is voluntary for both manufacturers and states. As of June 2026, seventeen major pharmaceutical companies—including industry giants like Pfizer and AstraZeneca—have signed MFN-related agreements. These companies have traded their commitment to lower pricing for a three-year reprieve from certain pharmaceutical tariffs. States, meanwhile, must opt into the program and commit to adopting uniform coverage criteria, such as standardized prior authorization and step therapy protocols, in exchange for these new supplemental rebates.
Chronology: A Timeline of Implementation
The rollout of the GENEROUS model has been marked by iterative adjustments to deadlines, reflecting the logistical challenges of integrating international pricing structures into the U.S. Medicaid framework.
- January 2026: The CMS Innovation Center officially launches the GENEROUS model with the stated goal of curbing rising prescription drug expenditures.
- March–April 2026: Recognizing the complexity for pharmaceutical firms to adjust their reporting and rebate structures, CMS extends the initial manufacturer application deadline twice, from March 31 to April 30, and eventually to June 11, 2026.
- April 2026: The White House releases a pivotal report estimating that a voluntary MFN framework could save the Medicaid program approximately $64.3 billion over the next decade.
- June 11, 2026: The final deadline for drug manufacturers to submit their applications to participate in the GENEROUS model.
- July 31, 2026: The deadline for individual states to submit their applications for participation.
- August 31, 2026: The deadline for states to finalize and execute formal participation agreements with CMS.
- 2026–2030: The five-year duration of the initial model, during which CMS will monitor and evaluate the impact of MFN pricing on program costs and patient access.
Supporting Data: The Landscape of Medicaid Spending
To understand the potential impact of GENEROUS, one must first look at how Medicaid currently handles drug spending. The existing Medicaid Drug Rebate Program (MDRP) is already a highly effective cost-containment tool.
The Efficiency of Current Rebates
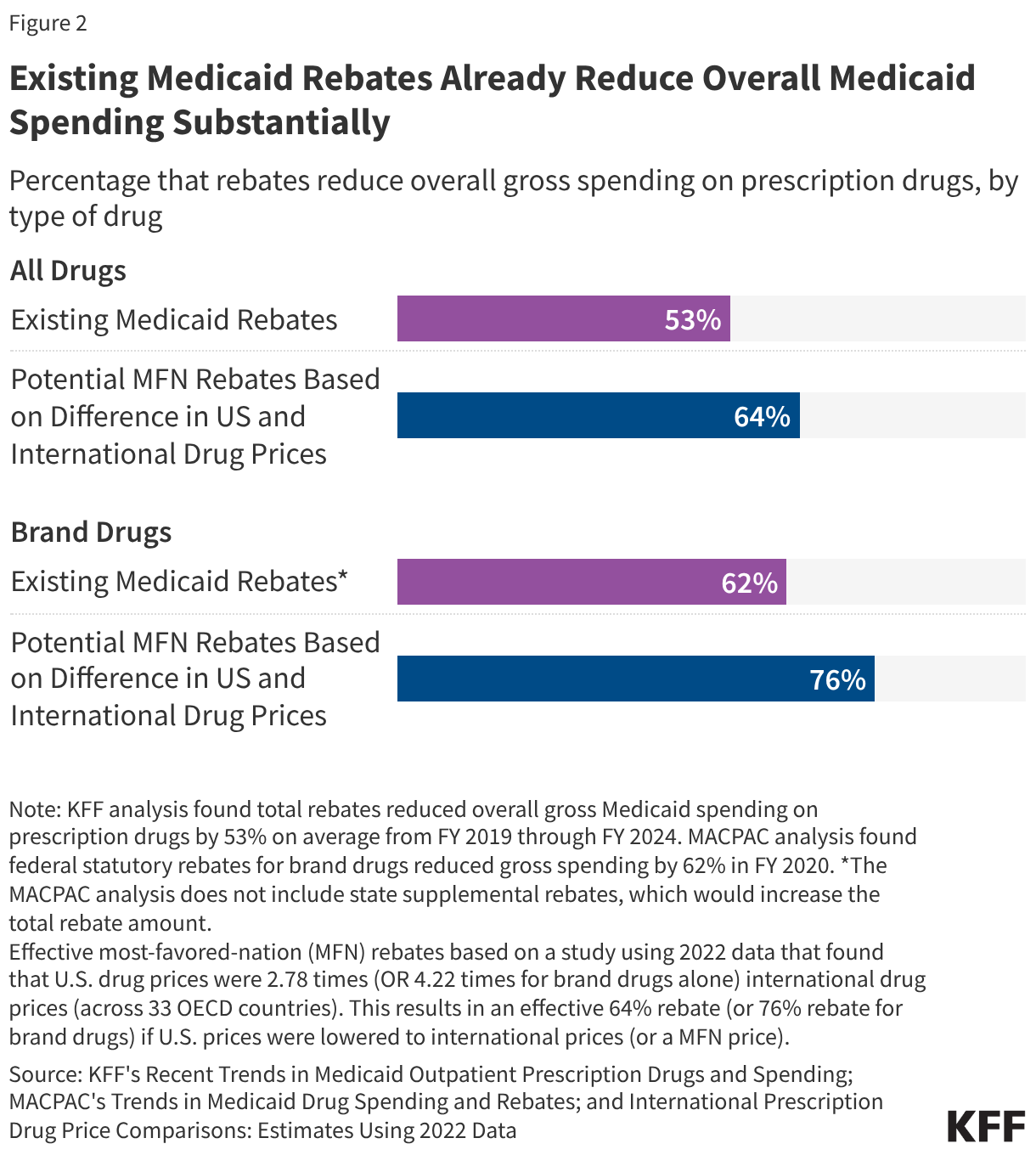
From fiscal year (FY) 2019 to FY 2024, statutory and supplemental rebates reduced gross Medicaid prescription drug spending by an average of 53%. For brand-name drugs, that figure is even higher, often exceeding 62%. Because of these deep existing discounts, the "room" for further savings through the MFN approach varies drastically by drug.

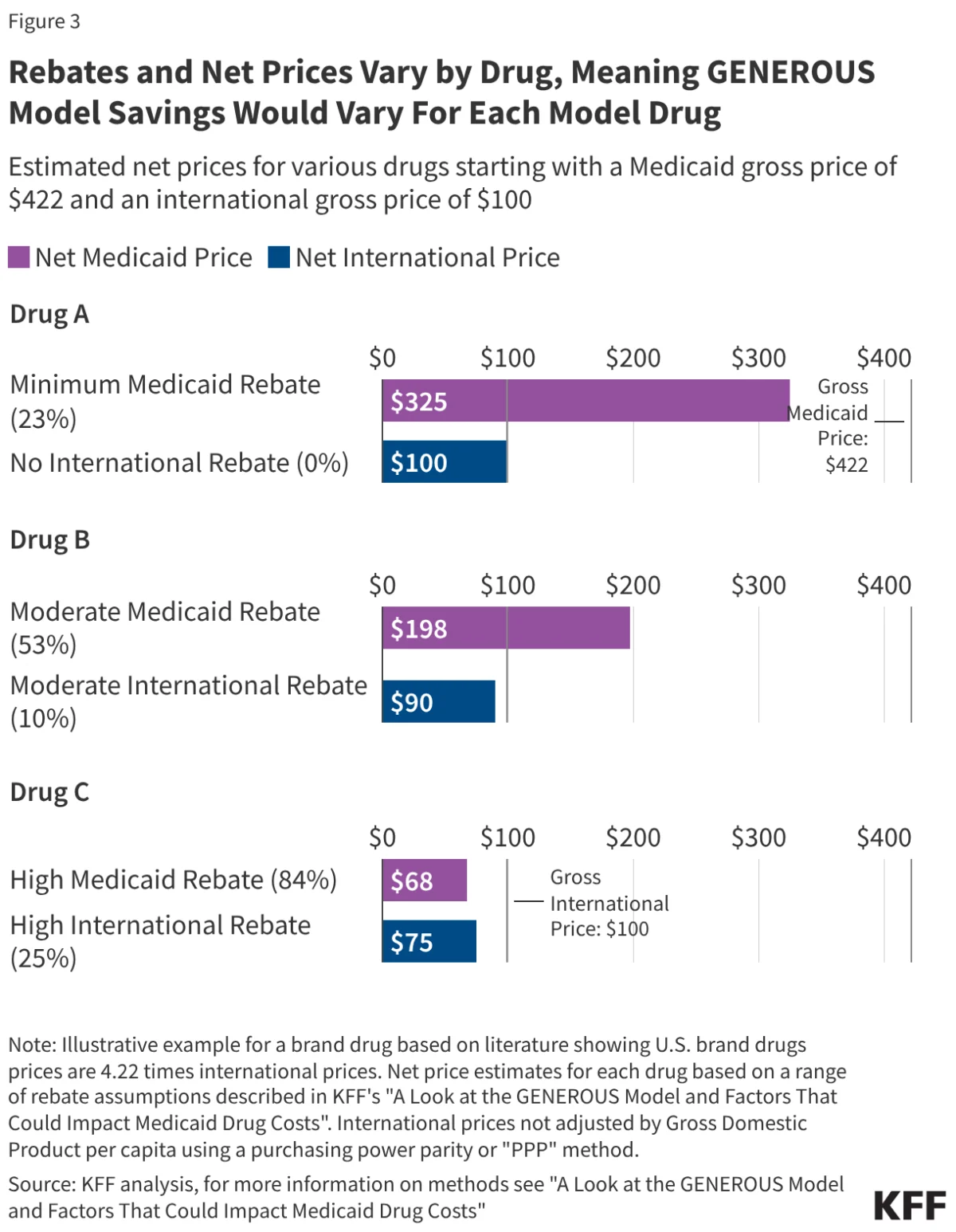
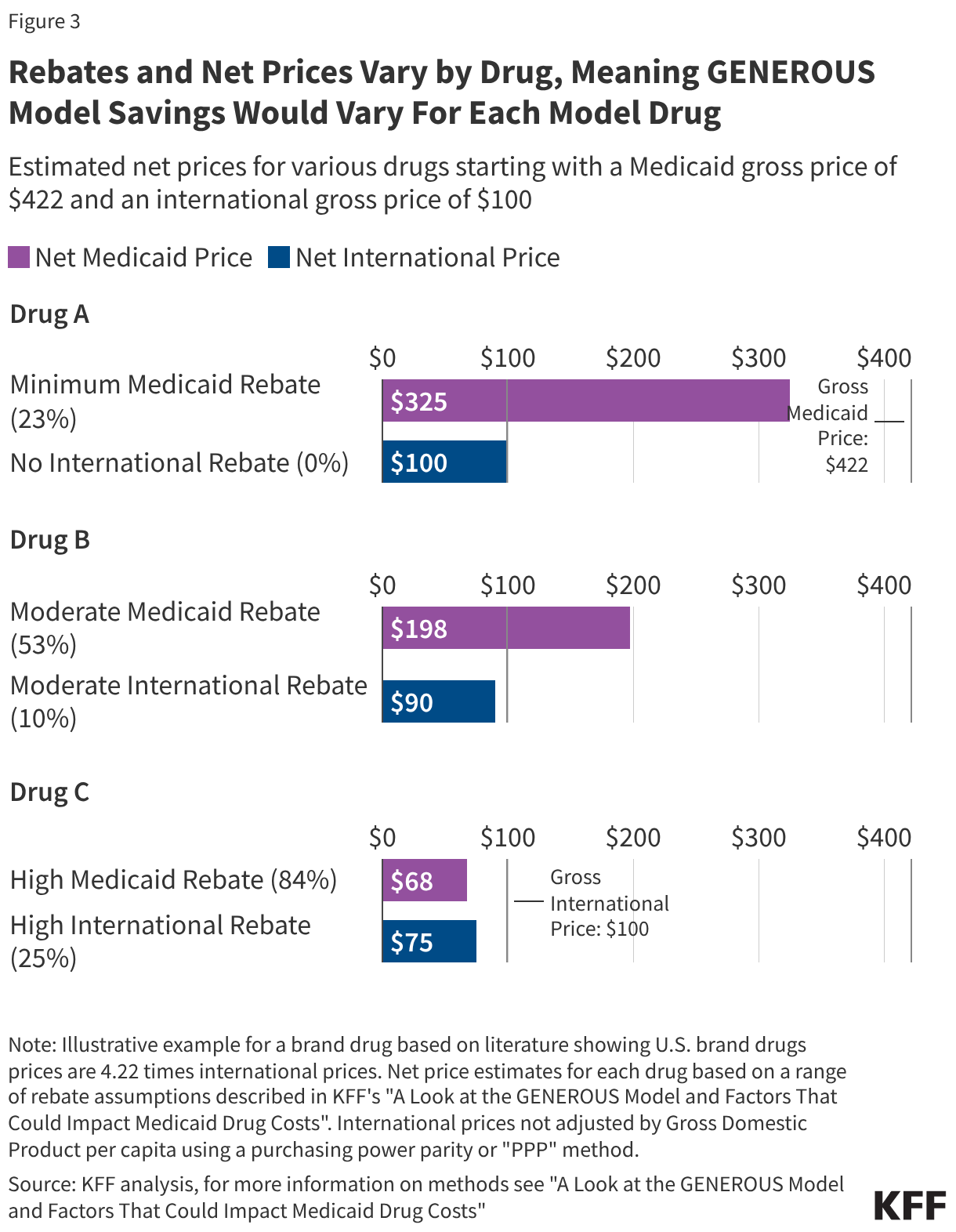
The "Drug A, B, and C" Dilemma
Analysis of current market dynamics reveals a split in potential savings:
- New/Niche Drugs (Drug A): Drugs with few competitors—often newer therapies—have smaller current rebates. The GENEROUS model is expected to generate the most significant savings here, as the gap between U.S. prices and international prices is widest.
- Mature/Generic-Competitive Drugs (Drug C): For drugs that have been on the market for a long time, states often already receive massive rebates (sometimes reaching 100% of the cost). In these instances, the GENEROUS model offers little additional relief, though it may provide administrative simplicity.
Furthermore, utilization is highly concentrated. The top 50 drugs account for over one-third of all Medicaid drug spending. Therefore, the success of the model hinges on whether these "high-spend" drugs are included in the MFN agreements.
Official Responses and Political Implications
The GENEROUS model has drawn sharp reactions from stakeholders across the political and economic spectrum.
Government and Legislative Scrutiny
The Trump administration champions the model as a necessary intervention against "price gouging" and a triumph of international trade leverage. However, the lack of transparency surrounding the specific terms of the agreements with companies like Pfizer has drawn concern from Congress. Senate leaders have issued formal letters to pharmaceutical executives, demanding evidence of cost savings to ensure that these secret deals actually benefit the taxpayer rather than simply serving as a temporary buffer against tariffs.
Pharmaceutical Industry Stance
For the pharmaceutical industry, the model is a calculated risk. While participating manufacturers face lower revenue per dose, they gain a three-year shield against looming pharmaceutical tariffs. Industry groups remain wary of the long-term precedent set by international price benchmarking, fearing that once these mechanisms are embedded, they will be difficult to dismantle.
State-Level Perspectives
States are currently conducting internal cost-benefit analyses. Because states already utilize diverse strategies—such as preferred drug lists (PDLs)—to negotiate supplemental rebates, many are hesitant to cede control to federal "uniform coverage criteria." If the federal criteria are more restrictive than what a state currently enforces, they could inadvertently limit patient access to necessary treatments. Consequently, state participation is expected to be highest in jurisdictions where current drug spending remains unmanaged and high.
Implications: What Does This Mean for the Future?
The long-term impact of the GENEROUS model remains speculative, yet several key implications are clear.
1. The Diminishing Returns of Savings
While the White House estimates average annual savings of $6.43 billion, these savings are likely to be front-loaded. As the U.S. market pushes for international pricing, global manufacturers may adjust their strategies, potentially raising prices in those reference countries to offset the losses incurred in the U.S. market. This "price leveling" could eventually erode the savings advantage of the GENEROUS model.
2. Administrative Efficiency vs. Clinical Flexibility
The shift toward standardized coverage criteria is a double-edged sword. On one hand, it reduces the administrative burden on states and manufacturers by creating a uniform playbook for prior authorization. On the other hand, it removes the ability of states to tailor their formularies to the specific needs of their unique Medicaid populations.
3. Impact on Innovation and Access
Critics of the MFN model argue that forcing price alignment with lower-cost nations may dampen investment in research and development (R&D). Conversely, proponents argue that the U.S. has subsidized global drug development for too long and that the GENEROUS model provides a necessary "market correction." For the Medicaid enrollee, the impact is expected to be minimal in terms of out-of-pocket costs, which are protected by federal law; however, the model could lead to shifts in which drugs are covered and how quickly new therapies are added to state formularies.
Conclusion
The GENEROUS model represents a significant evolution in the federal government’s approach to drug pricing. By attempting to import the price-negotiation power of foreign health systems, the Trump administration has challenged the status quo of the U.S. pharmaceutical market. Whether the model becomes a permanent fixture of Medicaid or a short-lived experiment will depend on the transparency of the current manufacturer deals, the willingness of states to trade autonomy for potential savings, and the ultimate ability of the model to prove that it can lower costs without sacrificing access to essential care. As the August 2026 deadline for state participation approaches, the pharmaceutical landscape in America stands at a critical, and still uncertain, crossroads.




