Breakthrough in Oncology: Disrupting the ELF3-HSP27 Interaction
Breast cancer remains the most frequently diagnosed malignancy and a leading cause of cancer-related mortality among women worldwide. Despite significant advancements in diagnostic techniques and therapeutic interventions, the high rates of recurrence and metastasis continue to pose a formidable challenge to clinical outcomes. A collaborative research team from Ewha Womans University and CHA University in South Korea has unveiled a critical molecular mechanism that drives this aggressive behavior, identifying a novel therapeutic target: the interaction between the transcription factor ELF3 and the molecular chaperone HSP27.
The study, published in the journal Pharmaceuticals, provides a detailed look at how these two proteins cooperate to fuel tumor growth and, more importantly, demonstrates the successful development of a small-molecule inhibitor, HT81, capable of disrupting this pathological partnership.

Chronology of the Discovery
The research process spanned several years, involving a rigorous combination of bioinformatics analysis, cellular engineering, and pharmacological screening:
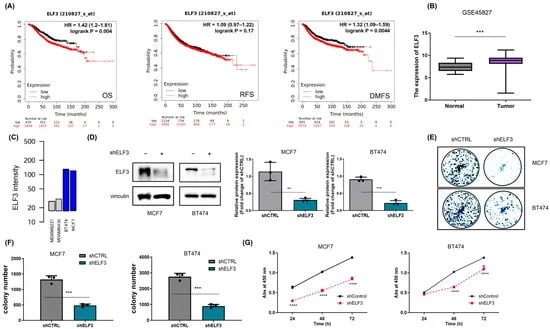
- May 2023: Researchers utilized the Kaplan-Meier Plotter and GEO datasets to confirm that elevated levels of ELF3 are significantly correlated with poor overall survival and distant metastasis in breast cancer patients.
- Late 2023–2024: Through a series of loss-of-function experiments, the team identified that silencing ELF3 effectively suppressed the proliferation of breast cancer cell lines like MCF7 and BT474.
- Early 2025: The team established that HSP27 (HSPB1) acts as a critical binding partner for ELF3. Using split luciferase complementation and GST pull-down assays, they mapped the interaction to the Transactivation Domain (TAD) of ELF3.
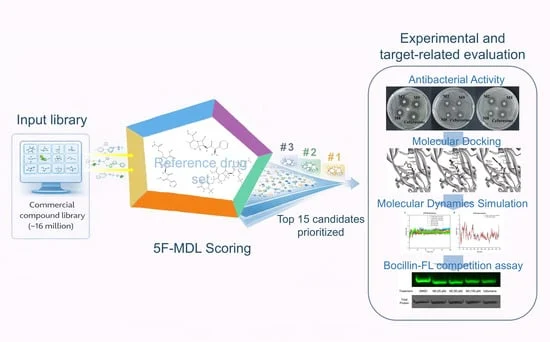
- Mid-2025: High-throughput screening of a library of 29 novel chalcone-derived compounds led to the identification of HT81, which demonstrated a potent ability to inhibit the ELF3-HSP27 binding interface.
- May 2026: Final validation in a BT474 xenograft mouse model confirmed that HT81 administration significantly reduced tumor volume and weight, leading to the formal publication of the study.
Supporting Data and Molecular Mechanisms
The efficacy of the research is rooted in a deep understanding of protein homeostasis. Heat shock proteins, particularly HSP27, are known for their role in protein folding and stability. The study reveals that HSP27 functions as a guardian for ELF3, preventing its degradation and facilitating its translocation into the cell nucleus.

Once inside the nucleus, ELF3 exerts its influence as a transcription factor, activating genes that promote rapid cell division and inhibit apoptosis. The researchers utilized a cycloheximide (CHX) chase assay to prove that when HSP27 is absent, the half-life of the ELF3 protein drops dramatically—from over 5 hours to less than 2 hours in certain cell lines.
The study’s data suggests that HT81 works by "unmasking" ELF3. By preventing HSP27 from binding to the TAD of ELF3, the compound destabilizes the transcription factor, causing it to undergo premature degradation. Consequently, the cancer cells lose their ability to sustain high-speed proliferation, leading to an increase in markers of cell death, such as cleaved PARP (C-PARP).

Official Perspectives and Scientific Significance
The lead authors, Youngjoo Kwon and Younghwa Na, emphasize that this research represents a shift from targeting individual proteins to targeting protein-protein interactions (PPIs). "Targeting the ELF3-HSP27 interaction enables us to modulate tumor-specific signaling networks with high precision," the authors noted in their discussion.
The research team highlighted that while many existing therapies target the protein itself, PPI inhibitors offer a more nuanced approach. By disrupting the complex, they can stop the oncogenic signal without necessarily deleting the protein entirely, which often results in fewer off-target toxicities. The institutional review board at Ewha Womans University approved all animal studies, which were conducted in strict adherence to the ARRIVE guidelines, ensuring that the results met the highest ethical and scientific standards.

Implications for Future Oncology
The discovery of HT81 as a lead compound carries significant implications for the future of breast cancer treatment:
A Personalized Approach to Therapy
The study observed that cells with high levels of both ELF3 and HSP27 were the most sensitive to HT81. In contrast, cell lines like MDA-MB-231, which express lower levels of these proteins, showed reduced sensitivity. This suggests that the HT81 treatment could be a "biomarker-driven" therapy, potentially benefiting patients whose tumors are specifically reliant on this chaperone-client axis.

Combating Treatment Resistance
A recurring theme in breast cancer mortality is the development of resistance to conventional chemotherapies. Because HSP27 is often upregulated in response to cellular stress, it frequently contributes to therapy resistance. By targeting the interaction between HSP27 and its client proteins, clinicians may be able to overcome resistance mechanisms, essentially "re-sensitizing" tumor cells to treatment.
The Path to Clinical Translation
While the results in xenograft models are highly encouraging—demonstrating significant tumor reduction at a dosage of 20 mg/kg/day—the authors are cautious about the next steps. They acknowledge that HT81 is a preliminary lead compound. Future research will focus on:

- Structural Optimization: Improving the pharmacological properties and potency of HT81 analogs.
- Target Engagement: Clarifying the exact binding site of HT81 on the protein complex to refine its specificity.
- Broadened Scope: Testing the inhibitor against other aggressive breast cancer subtypes, including Triple-Negative Breast Cancer (TNBC), to determine the full therapeutic window of this strategy.
In conclusion, the study provides a robust scientific rationale for investigating PPIs in oncology. By proving that the ELF3-HSP27 axis is a viable target, the researchers have opened a new pathway for drug development that could eventually lead to more effective, less toxic, and highly targeted treatments for breast cancer patients. As the research moves toward potential clinical trials, the focus will remain on refining these small-molecule inhibitors to ensure they can safely and effectively navigate the complex landscape of human tumor biology.