Main Facts: Solving a Diagnostic Dilemma
Langerhans cell sarcoma (LCS) is an exceptionally rare, highly aggressive malignant tumor derived from Langerhans cells. Because it shares the same cellular origin as the more common—and generally less malignant—Langerhans cell histiocytosis (LCH), clinicians have long struggled to differentiate the two in a pathological setting. This diagnostic ambiguity often leads to delays in identifying the life-threatening nature of LCS, which carries a significantly poorer prognosis.
A recent study published in the journal Diagnostics has introduced a potential paradigm shift in this field. Researchers have identified that the expression patterns of the Apoptosis-Stimulating Proteins of p53 (ASPP) family—specifically ASPP2 and iASPP—can serve as reliable immunohistochemical markers to distinguish LCS from LCH. Furthermore, the study reports the first-ever discovery of a specific MAP2K1 gene mutation (c.607G > A) in LCS, providing a new genetic target for molecular diagnostics and future therapeutic exploration.

Chronology: From Clinical Observation to Molecular Discovery
The research, led by Changsong Wang, Junling An, and colleagues at the 989th Hospital of the PLA Joint Logistic Support Force, spanned from January 2006 to March 2023. The study followed a rigorous multi-stage investigative process:
- Initial Collection (2006–2023): The team compiled clinical data and tissue samples from six patients diagnosed with LCS and six with LCH. All participants provided informed consent, and the study received ethical approval from the hospital’s review board.
- Histopathological & Immunohistochemical Screening: Researchers performed H&E staining to analyze cellular morphology and used immunohistochemistry (IHC) to test for the expression of ASPP family members (ASPP1, ASPP2, and iASPP).
- Molecular Validation: Using the amplification refractory mutation system (ARMS), the team screened for common genetic drivers, including BRAF V600E, K-ras, and ROS1.
- Whole-Exon Sequencing (WES): Recognizing that common markers were absent in many cases, the team utilized WES to identify novel genomic mutations in the six LCS samples.
- Publication (May 2026): The findings were formally published on May 7, 2026, establishing a new framework for clinical differentiation between these two conditions.
Supporting Data: The ASPP and MAP2K1 Connection
The study’s findings provide a clear visual and genetic roadmap for pathologists.

The ASPP Family Profile
The research found that the ASPP family members act differently in LCH compared to LCS:
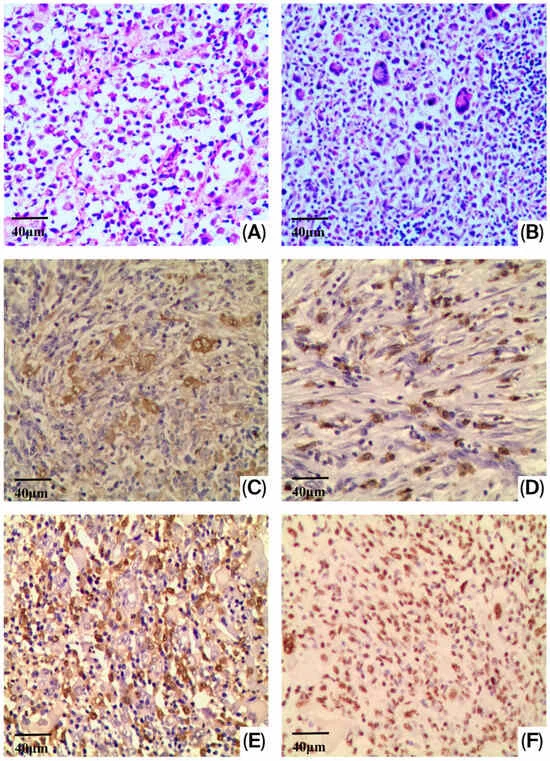
- ASPP1: Found to be absent in both LCH and LCS, rendering it ineffective as a diagnostic marker.
- ASPP2: In LCH, ASPP2 was present in both proliferating Langerhans cells and surrounding histiocytic cells. However, in LCS, it was exclusively found in the nuclei of ovoid and elliptic tumor cells.
- iASPP: This protein served as a major differentiator. It was expressed in the cytoplasm of tumor cells in 83% (5/6) of LCS cases, whereas no expression was observed in LCH.
Triple-color immunofluorescence confirmed that these proteins co-localize with Langerin, the standard marker for Langerhans cells, validating that the expression is intrinsic to the malignant cells themselves.

Genetic Findings
The team’s genetic analysis yielded a significant breakthrough. While previous studies have often looked for BRAF V600E mutations (common in LCH), this study found no such mutations in the LCS samples. Instead, the researchers identified a somatic driver mutation: MAP2K1 c.607G > A. This specific mutation results in an amino acid substitution from Glutamic acid (E) to Lysine (K) at the 203 site, occurring in 66.67% of the LCS cases studied. This discovery represents the first time this specific genetic variant has been documented as a potential marker for LCS.
Official Responses and Clinical Context
In their discussion, the authors highlighted the severity of LCS, noting that with a mortality rate of approximately 50% and many patients succumbing within 18 months, the need for rapid, accurate diagnosis is critical.

"The differential diagnosis of LCS and LCH is challenging as both share the same cell origin and similar histological features," the researchers stated. While previous studies have suggested markers like WT1 and CD44, or the viral load of Merkel cell polyomavirus, the diagnostic difficulty remains high in clinical practice. The research team emphasizes that by integrating the ASPP expression pattern with the identification of the MAP2K1 c.607G > A mutation, clinicians can move toward a more objective diagnostic protocol.
Implications for Future Oncology
The implications of this study are threefold:

- Diagnostic Precision: Pathologists now have a standardized immunohistochemical panel (ASPP2 and iASPP) that can be easily integrated into routine laboratory workflows, allowing for faster differentiation between the two conditions.
- Therapeutic Targeting: The identification of the MAP2K1 mutation provides a potential pathway for targeted therapy. Because MAP2K1 (MEK1) mutations are susceptible to MEK inhibitors like trametinib—which have already been used in other forms of histiocytosis—this finding could lead to off-label or clinical trial-based treatment options for LCS patients.
- Future Research Directions: The authors acknowledge that the small sample size is a limitation. They have called for larger, multi-center studies and the use of spatial omics to further validate these findings. As technology advances, the researchers plan to expand their molecular detection methods to ensure that these markers can be used with high confidence in a clinical setting.
In summary, this research from the 989th Hospital represents a significant leap forward in understanding the molecular landscape of Langerhans cell sarcoma. By moving beyond traditional histological examination and into the realm of specific protein expression and genomic mutation analysis, the medical community is now better equipped to diagnose and potentially treat one of the rarest and most aggressive tumors in the field of pathology.
Glossary of Key Terms
- Langerhans Cell Sarcoma (LCS): A rare and highly malignant tumor originating from Langerhans cells.
- Langerhans Cell Histiocytosis (LCH): A disorder characterized by the proliferation of Langerhans cells, typically less aggressive than LCS.
- ASPP Family: Proteins that regulate the tumor-suppressor p53, playing a vital role in cell apoptosis (programmed cell death).
- MAP2K1: A gene that encodes the MEK1 protein; mutations in this gene are associated with various cancers and immune disorders.
- Whole-Exon Sequencing (WES): A genomic technique for sequencing all the protein-coding regions of genes in a genome.
- Immunohistochemical (IHC) Method: A laboratory technique that uses antibodies to detect specific proteins in tissue samples.




